ORAL MICROBIOLOGY
Overview
The mouth is colonized by microorganisms a few hours after birth, mainly by aerobic and facultative anaerobic organisms. The eruption of teeth allows the development of a complex ecosystem of microorganisms (>300 species have been identified) and the healthy mouth depends on maintaining an environment in which these organisms coexist without damaging oral structures.
Microorganisms worth noting
Streptococcus mutans group. Several species are recognized within this group, including S. mutans and S. sobrinus. Aerobic. Synthesizes dextrans. Colony density rises to >50% in presence of high dietary sucrose. able to produce acid from most sugars. Most important organisms in the aetiology of caries.
Streptococcus oralis group includes S. sanguis, S. mitis, and S. oralis. Account for up to 50% of streptococci in plaque. Heavily implicated in 50% of cases of infective endocarditis.
Streptococcus salivarius group. Accounts for about half the streptococci in saliva. Inconsistent producer of dextran.
S. intermedius, S. angiosus, S. constellatus (formerly S. milleri group). Common isolates from abscesses in the mouth and at distant sites.
Lactobacillus. Secondary colonizer in caries. Very acidogenic. Often found in dentine caries.
Porphyromonas gingivalis. Obligate anaerobe associated with chronic periodontitis and aggressive periodontitis.
Prevotella intermedia. Found in chronic periodontitis, localized aggressive periodontitis (juvenile periodontitis), necrotizing periodontal disease, and areas of severe gingival inflammation without attachment loss.
Prevotella nigrescens. New, possibly more virulent.
Fusobacterium. Obligate anaerobes. Originally thought to be principal pathogens in necrotizing periodontal disease. Remain a significant periodontal pathogen.
Borrelia vincenti (refringens). Large oral spirochaete; probably only a co-pathogen.
Actinobacillus actinomycetemcomitans. Microaerophilic, capnophilic. Gram negative rod. Particular pathogen in juvenile periodontitis and rapidly progressive periodontitis.
Actinomyces israeli. Filamentous organism; major cause of actinomycosis. A persistent rare infection which occurs predominantly in the mouth and jaws and the female reproductive tract. Implicated in root caries.
Candida albicans. Yeast-like fungus, famous as an opportunistic oral pathogen; probably carried as a commensal by most people.
Spirochaetes. Obligate anaerobes implicated in periodontal disease; present in most adult mouths. Borrelia, Treponema, and Leptospira belong to this family.
PLAQUE
Dental plaque, which is a biofilm, is a firmly adherent mass of bacteria in a muco-polysaccharide matrix. It cannot be rinsed off but can be removed by brushing. It is the root of most dental evils.
Attachment. Although it is possible for plaque to collect on irregular surfaces in the mouth, to colonize smooth tooth surfaces it needs the presence of acquired pellicle. This is a thin layer of salivary glycoproteins, formed on the tooth surface within minutes of polishing. The pellicle has an ion-regulation function between tooth and saliva and contains immuno-globulins, complement, and lysozyme.
Development. Up to 106 viable bacteria per mm2 of tooth surface can be recovered 1 h after cleaning; these are selectively adsorbed streptococci. Bacteria recolonize the tooth surface in a predictable sequence. Streptococcus mutans synthetizes extracellular polysaccharides (glucan and fructan) specifically from sucrose and promotes its early colonization in this way. Cocci predominate in plaque for the first 2 days, following which rods and filamentous organisms become involved. This is associated with ↑ numbers of leucocytes at the gingival margin. Between 6 and 10 days, if no cleaning has taken place, vibrios and spirochaetes appear in plaque and this is associated with clinical gingivitis. It is generally felt that the move towards a more Gram negative anaerobe-dense plaque is associated with the progression of gingivitis and periodontal disease.
Plaque in caries. As several oral streptococci, most notably mutans streptococci, secrete acids and the matrix component of plaque, there is a clear relationship between the two. However, various other factors complicate the picture, including saliva, other microorganisms, and the structure of the tooth surface.
Plaque in periodontal disease. There is a direct correlation between the amount of plaque at the cervical margin of teeth and the severity of gingivitis, and experimental gingivitis can be produced and abolished by suspending and reintroducing oral hygiene. It is commonly accepted that plaque accumulation causes gingivitis, the major variable being host susceptibility. While there are numerous interacting components which determine the progression of chronic gingivitis to periodontitis, particularly host susceptibility, the presence of plaque, particularly 'old' plaque with its high anaerobe content, is widely held to be crucial, and most Rx is based on the meticulous, regular removal of plaque.
CALCULUS
Calculus (tartar) is a calcified deposit found on teeth (and other solid oral structures) and is formed by mineralization of plaque deposits. It can be subdivided into:
Supragingival calculus, most often found opposite the openings of the salivary ducts, i.e. 76|67 opposite the parotid (Stensen's) duct and on the lingual surface of the lower anterior teeth opposite the submandibular / sublingual (Wharton's) duct. It is usually yellow, but can become stained a variety of colours.
Subgingival calculus is found, not surprisingly, underneath the gingival margin and is firmly attached to tooth roots. It tends to be brown or black, is extremely tenacious, and is most often found on interproximal and lingual surfaces. It may be identified visually, by touch using a WHO 621 probe, or on radiographs. With gingival recession it can become supragingival.
Composition. Consists of up to 80% inorganic salts, mostly crystalline, the major components being calcium and phosphorus. The microscopic structure is basically that of a randomly orientated crystal formation.
Formation is always preceded by plaque deposition, the plaque serving as an organic matrix for subsequent mineralization. Initially, the matrix between organisms becomes calcified with, eventually, the organisms themselves becoming mineralized. Subgingival calculus usually takes many months to form, whereas friable supragingival calculus may form within 2 weeks.
Pathological effects. Calculus (particularly, subgingival calculus), is associated with periodontal disease. This may be because it is invariably covered by a layer of plaque. Its principal detrimental effect is probably that it acts as a retention site for plaque and bacterial toxins. The presence of calculus makes it difficult to implement adequate oral hygiene.
AETIOLOGY OF PERIODONTAL DISEASE
Overview
Plaque is the principal aetiological factor in virtually all forms of periodontal disease. Periodontal damage is almost certainly the direct consequence of colonization of the gingival sulcus by organisms within dental plaque. However, the progression from gingivitis to periodontitis is far more complex than this statement suggests, as it involves host defence, the oral environment, the pathogenicity of organisms, and plaque maturity. It is probably easiest to regard periodontal disease as a complex multifactorial infection complicated by the inflammatory response of the host. Various elements of this process are worthy of special note:
Microbiology
The changing microbiology of dental plaque has already been referred to. The inflammatory response of gingiva to the presence of initial young plaque creates a minute gingival pocket which serves as an ideal environment for further bacterial colonization, providing all the nutrients required for the growth of numerous fastidious organisms. In addition, there is an extremely low oxygen level within gingival pockets, which favours the development of obligate anaerobes, several of which are closely associated with the progression of periodontal disease. High levels of carbone dioxide favour the establishment of capnophilic organisms, some of which are associated with localized aggressive periodontitis (LAP).
Briefly, clinically healthy gingivae are associated with a high proportion of Gram positive rods and cocci which are facultatively anaerobic or aerobic. Gingivitis is associated with ↑ number of facultative anaerobes, strict anaerobes, and an increasing number of Gram negative rods. Established periodontitis is associated with a majority presence of anaerobic Gram negative rods. Specific organisms involved in periodontal disease worthy of note include: Porphyromonas gingivalis, Prevotella intermedia P. denticola (previously Bacteroides), and spirochaetes. They have many properties which ↑ pathogenicity, particularly activity against neutrophils. Actinobacillus actinomycetemcomitans, a capnophilic organism thought to be involved in the aetiology of LAP, is also active against neutrophils. However, to date it has not been possible to identify one particular organism or group of microorganisms solely responsible for the initiation and progression of periodontal disease, although the general concepts outlined reflect current working data.
Immunopathology
The inflammatory response to the presence of dental plaque is detectable both clinically and histologically, and is certainly responsible for at least some of the periodontal destruction which occurs. Both inflammatory and immunologically mediated pathways can contribute to periodontal damage. Antigenic substances released by plaque organisms elicit both cell-mediated and humoral responsess which, while designed to be protective, also cause local tissue damage, usually by complement activation (Bystander Damage). Non-immune mediated damage is caused by one or all of the major endogenous mediators of inflammation: vasoactive amines (histamine), plasma proteases (complement), prostaglandins and leukotrienes, lysosomal acid hydrolases, proteases, free radicals, and cytokines.
Host
Local and systemic modifying factors influence progress of the disease.
Systemic factors include immune status, stress, endocrine function (e. g. diabetes), smoking, drugs, age, and nutrition. There has been evidence reported suggesting a link between periodontal, ischaemic heart, and cerebrovascular disease. It is unclear if this is a cause and effect relationship. Watch this space.
Local factors are tooth position and morphology, calculus, overhangs and appliances, occlusal trauma, and mucogingival state.
EPIDEMIOLOGY OF PERIODONTAL DISEASE - 1
Epidemiology is the study of the presence and effect of disease on a population. In order for this to be of value it is essential to be able to quantify the prevalence and degree of severity of any given disease, in a reproducible manner. The search for suitable indices in periodontology has left the literature replete with often confusing and redundant scoring systems.
The original purpose of periodontal indices was to study the extent of disease within population groups; however, the value of indices in the screening and management of individual patients soon became apparent. The following two indices fulfil both these criteria and are simple and easy to perform:
Debris or Oral Hygiene Index. This can be modified for personal use by using disclosing agents.
0 No debris or stain.
1 Soft debris covering not more than 1/3 of the tooth surface.
2 Soft debris covering more than 1/3 but less than 2/3.
3 Soft debris covering over 2/3 of tooth surface.
Basic Periodontal Examination (BPE). Also known as Community Periodontal Index of Treatment Needs (CPITN). This technique is used to screen for those patients requiring more detailed periodontal examination. It examines every tooth in the mouth (except third molars), thus taking into acount the site-specific nature of periodontal disease. A World Health Organization (WHO) periodontal probe (ball-ended with a coloured band 3,5-5,5 mm from the tip) should be used. The mouth is divided into sextants, i. e. two buccal and one labial segment per arch. Six sites on each tooth and the highest score per sextant recorded, usually in a simple six-box chart.
0 = No disease,
1 = Gingival bleeding but no pockets, no calcuclus, no overhanging restoration. Rx: OHI.
2 = No pockets > 3 mm, subgingival calculus present or subgingival retention site, e. g. overhang. Rx: OHI, scaling, and correction of any iatrogenic problems.
3 = Deepest pocket 4 or 5 mm. Rx: OHI, scaling, and root planning.
4 = One or more tooth in sextant has a pocket > 6 mm. Rx: scaling and root planning, and/or flap as required.
* = Furcation or total loss of attachment of 7 mm or more. Rx: full periodontal examnation of the sextant regardless of CPITN score.
Individuals identified as having areas of advanced periodontal disease will require a full probing depth chart, together with recordings of mobility, recession and furcation involvement, and radiological examination. BPE cannot be used for close monitoring of the progress of Rx.
EPIDEMIOLOGY OF PERIODONTAL DISEASE - 2
Other techniques for assessing the levels of periodontal disease include:
Marginal bleeding index (MBI). Score 1 or 0 depending on whether or not bleeding occurs after a probe is gently run around the gingival sulcus. A percentage store is obtained by dividing by the number of teeth and multiplying the result by 100.
Plaque index (PlI). This is based on the presence or absence of plaque on the mesial, distal, lingual, and buccal surfaces revealed by disclosing.
Percentage score = (number of surfaces with plaque x 100 ) / (total number of teeth x 4)
Both the MBI and PlI can be expressed as bleeding or plaque-free scores in this way obtaining a high score is a good thing, which may be both easier for the patient to understand and a more positive motivational approach.
Although periodontal diseases are very common, severe forms affect no more than about 10-15% of the population. Overall, periodontitis accounts for between 30 and 35% of all tooth extractions; caries and its consequences account for up to 50%.
The direct association between the presence of tooth surface plaque and periodontal damage has been confirmed, but the rate of destruction has been shown to vary, not only between individuals but also between different sites in the same mouth, and at different times in the same individual.
WHO probe for use in BPE/CPITN
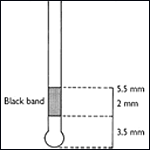
If black band disappears in probing pocket, perform full periodontal examination in that sextant.
CHRONIC GINGIVITIS
Chronic gingivitis is, as the name suggests, inflammation of the gingival tissues. It is not associated with alveolar bone resorption or apical migration of the junctional epithelium. Pockets > 2 mm can occur in chronic gingivitis due to an increase in gingival size because of oedema or hyperplasia (false pockets). Four different types of gingivitis are described; the commonest type is plaque induced.
Plaque-induced ginvigitis. This is present in virtually all mouths to some extent. The classic triad of redness, swelling, and bleeding on gentle probing are diagnostic and are usually associated with a complaint by the patient that their'"gums bleed on brushing'. False pocketing may also be present. This gingivitis occurs as a result of low-grade infection caused by the presence of undisturbed dental plaque, which is associated with a change in the flora from Gram positive aerobes to Gram negative anaerobes. This gives rise to inflammatory changes in the associated gingivae; these are detectable histologically prior to the appearance of overt clinical gingivitis, which is observed after about 7 days of undisturbed plaque accumulation. The inflammatory response seen comprises an alteration in the integrity of the gingival microcirculation, an ↑ in the numbers of inflammatory cells in the gingival connective tissue (i. e. plasma cells, lymphocytes, macrophages, and neutrophils), a ↑ in the number of fibroblasts, and a ↑ in collagen density. These inflammatory changes are easily reversible after institution of effective plaque control. While gingivitis is reversible, it should be remembered that calculus and other factors which promote plaque retention (e. g. overhanging restorations) will make adequate oral hygiene difficult. These factors should therefore be corrected by scaling and appropriate restorative treatment in addition to OHI. Gingivitis may be a precursor to, or marker of, adult periodontitis, and this must be excluded by measuring probing attachment levels (looking for 'true' pockets) and excluding the presence of alveolar bone loss by radiographs, if indicated. Other forms of gingivitis include:
Gingivitis modified by systemic factors. These would include puberty-associated gingivitis, menstrual cycle-associated gingivitis, pregnancy-associated gingivitis, pyogenic granuloma, diabetes mellitus-associated gingivitis, and gingivitis associated with blood dyscrasias, e. g. leukaemia-associated gingivitis.
Gingivitis modified by medications. These would include drug-influenced gingival enlargement and drug-induced gingivitis, e. g. oral contraceptive-associated gingivitis and drug-induced gingival overgrowth due to phenytoin or cyclosporin.
Gingival disease modified by malnutrition. These would include ascorbic acid-deficiency gingivitis (scurvy) and gingivitis due to protein deficiency.
CLASSIFICATION OF PERIODONTAL DISEASE
There appears to be a deep need among the 'labellers' to rename and reclassify microorganisms and diseases. Periodontitis has suffered more than most from this.
In truth, the only real benefit your patient receives from this exercise is if the classification has a bearing on Rx, and fortunately this is something that is starting to happen among periodontologists.
There are, of course, the usual caveats about patients being individuals and the real difficulty in making highly specific diagnosis in individual cases. Despite this, the current classification may prove helpful in thinking your way through the different periodontal diseases:
I Chronic periodontitis.
II Aggressive periodontitis.
III Periodontitis as a manifestation of systemic disease.
IV Necrotizing periodontal diseases.
V Abscesses of the periodontium.
VI Periodontitis associated with endodontic lesions.
VII Development or acquired deformities and conditions.
CHRONIC ADULT PERIODONTITIS
Chronic periodontitis (CP) can be regarded as a progression of the combination of infection and inflammation of gingivitis into the deep tissues of the periodontal membrane. It is characterized by breakdown of periodontal fibre bundles at the cervical margin, resorption of alveolar bone, and apical proliferation of junctional epithelium beyond the amelocemental junction. The progression of chronic gingivitis to periodontitis is by no means straightforward, as it varies in rate and progression not only between individuals, but also berween sites within the same mouth, and with time. It is now thought that periodontal destruction occurs in acute bursts of disease activity, each lasts for a variable period of time and is probably induced by a change in the quantity and/or quality of the subgingival microflora in undisturbed plaque, although other parameters may be involved. These include host response due to systemic disease, and local factors such as occlusal trauma, iatrogenic damage, or an as yet undetermined variable in the inflammatory response. The quiescent phase is associated with no advance in disease on either clinical or radiographic grounds and may last for extended periods of time. However, complete healing of the lesion does not occur because plaque remains on the root surface, and inflammation therefore persists in the connective tissues.
Microbiology. In the majority of cases the microbial pathogens remain within the plaque in the periodontal pocket and do not invade the periodontal tissues. It has already been mentioned that sampling of pockets in cases of CP consistently recovers a greatly ↑ proportion of Gram negative anaerobic rods and spirochaetes. Debate still continues between adherents of the specific (CP caused by specific organisms), and the non-specific plaque (CP caused by quantity rather than quality of plaque) theories. Nevertheless, certain Gram-negative anaerobes appear particularly prominently in active disease sites; namely Porphyromonas gingivalis, Prevotella intermedia/nigrescens, Bacteroides forsythus, Campylobacter rectus, and Treponema denticola (apologies to the reader, microbiologists appear to have a pathological need to rename organisms). Some of these organisms synthesize enzymes such as proteases which would exert a deleterious effect on the periodontal membrane, and all Gram negative organisms release endotoxin (their cell wall) on death.
Diagnosis is based on:
- Probing to elicit bleeding (which is the single most useful indicator of disease activity), measuring pocket depth attachment levels, and detecting subgingival calculus.
- Testing teeth for mobility and vitality.
- Radiographic examination (vertical bitewings and periapicals). See radiological selection criteria.
Treatment: below.
Generally aggressive periodontitis is a severe form of generalized periodontitis affecting young adults (20-35 yrs). Affects 1-2% of the Western population with an ↑ in Afro-Caribbeans. Microbiologically -- round-up the usual suspects plus A. actinomycetemcomitans. Immunologically there may be an associated neutrophil defect. Rx as for LAP.
Refractory periodontitis is characterized by low plaque scores and a poor response to appropriate periodontal therapy; 4-8% of patients with CP have refractory periodontitis. >90% are smokers.
POCKETING
Periodontal pockets can be divided into the following:
False pockets are due to gingival enlargement with the pocket epithelium at or above the amelocemental junction.
True pockets imply apical migration of the junctional epithelium beyond the amelocemental junction and can be divided into suprabony and infrabony pockets. Infrabony are described according to the number of bony walls: three-walled defect is the most favourable, as it is surrounded on three sides by cancellous bone and on one side by the cementum of the root surface. Two-walled defect may be either a crater between teeth having bone on two walls and cementum on the other two, or have two bony walls, the root cementum, and an open aspect to the overlying soft tissues. One-walled defects may be hemiseptal through-and-through defects, or one bony wall, two root cementum, and one soft tissue.
Pocket depths are measured from the gingival margin to the estimated base of the pocket. Attachment levels are measured from a fixed reference point: the cement-enamel junction or margin of a restoration to the base of the pocket. Pockets are therefore dependent on the position of the gingival margin.
Periodontal probes are the key instruments in detecting pockets. Numerous designs exist, and while individual preference will influence choice, it is sensible to reduce variability by selecting a single type of probe and using that type of probe throughout any one individual's Rx. The use of the WHO probe for screening using the BPE index is described above. Patients who are identified as having advanced CP should then be investigated further, including probing around each tooth. The main other indicator of periodontal disease, bleeding, is also detected using a probe (gently), and again consistency with a single type of probe is necessary.
Probing variables. The depth of penetration depends upon:
- Type of probe and its position.
- Amount of pressure used.
- Degree of inflammation.
It is now apparent that the measurement obtained with a probe does not correspond to sulcus or pocket depth. In the presence of inflammation a probe tip can pass through the inflamed tissues until it reaches the most coronal dento-gingival fibres, about 0,5 mm apical to the apical extent of the junctional epithelium, i. e. an overestimation of the problem. The amount of penetration into the tissues varies directly with the degree of inflammation, so that, following resolution of inflammation, an underestimate of attachment levels may be given. Formation of a tight, long junctional epithelium following Rx may also give a false sense of security if probing measurements are not interpreted with a degree of caution. For this reason the term 'probing pocket depth' is preferred to pocket depth.
DIAGNOSTIC TESTS AND MONITORING
It is widely accepted that disease active and inactive pockets exist. Progression is episodic and more likely in susceptible patients. Bleeding on probing has traditionally been the most useful indicator of disease activity; however, only 30% of sites which bleed will go on to lose attachment.
With ↑ emphasis on specific periodontopathic bacteria and availability of assays for components of immunological response, chairside diagnostic tests using gingival crevicular fluid have been developed.
Three types exist:
- Based on detection of antibodies to specific periodontopaths, e. g. Kodak Evalusite.
- Based on components of immune response, e. g. elastase in Dentsply Prognostick.
-
Based on tissue breakdown by products, e. g. aspartate aminotransferase in Colgate-Palmolive's Periogard.
These aim to predict sites of future and actual disease progression, and the first may indicate need for specific antibiotic therapy.
There is a huge amount of ongoing research into improving and refining these tests, but evidence is still required to demonstratve predictive ability and a higher level of accuracy than bleeding on probing.
Other peripheral techniques worthy of note are pocket temperature probes and computerized subtraction radiovisiography.
Radiographs are useful in comparing degree of bone loss and root surface deposits with pocket depth. Standardized sequential radiographs allow monitoring of disease.
-
Horizontal bitewings provide a good view of interproximal bone, useful for relatively minor degrees of bone loss (pocketing <5 mm) and to detect calculus deposits. Vertical bitewings are recommended when pocketing is >5 mm.
-
Full mouth periapicals (long cone technique), supplemented with vertical or horizontal bitewing, have been the radiological assessment of choice for patients with significant periodontal disease, i. e. irregular pocketing. They can clearly demonstrate root surface deposits, furcation involvement, extensive bone loss, infrabony pocketing, and perio-endo lesions. Radiation exposure is > )PG but it gives a more accurate representation of bone loss.
The results of a radiographic examination, clinical assessment, and assessment of pocket depth can all be marked on an updatable periodontal chart to monitor progress with Rx.
ACUTE PERIODONTAL DISEASE
Necrotizing ulcerative gingivitis (NUG) is also known as Vincent's gingivitis, Vincent's gingivostomatitis, ulceromembraneous gingivitis, or trench mouth. It should not be confused with Vincent's angina, which is also a fusospirochaetal infection but is typically localized over the tonsils.
NUG is characterized by painful papillary yellowish-white ulcers which bleed readily. Patients also often complain of a metallic taste and the sensation of their teeth being wedged apart. Regional lymphadenitis, fever, and malaise may occur in some cases. NUG is associated with poor oral hygiene, but stress and smoking act as cofactors. Inadequately treated NUG will lapse into a less symptomatic form which has become known as chronic ulcerative gingivitis, in which a slower rate of destruction occurs. NUG is usually a limited gingival condition, but a rare and more serious form known as cancrum oris or noma is found in patients who are malnourished, and in this form can lead to extensive destruction of the jaws and face.
Microbiology. Originally Borrelia vincenti (refringens) and Fusobacterium fusiformis were held to be
the major culprits in a mixed anaerobic infection. Further research has, however, served to confuse
the picture, with Porphyromonas spp. being implicated, as well as Treponema spp. Selenomonas
spp. and Prevotella spp. The crucial aspect of the microbiology of NUG is that it is a Gram negative
anaerobic infection which has been shown to actually invade the tissues but usually responds to
local debridement. In recent years, the tendency for patients who are HIV positive to develop
severe NUG, sometimes with bone necrosis, has renewed interest in it. From the practical point of
view it should be remembered that NUG presenting in an otherwise apparently healthy young adult
may be a presenting sign of HIV infection. Practically, consider control of cross-infection, examining
the mouth for other signs of infection, and directing the patient to appropriate counselling
and/or HIV testing.
Rx. In most cases local measures, i.e. thorough debridement and adequate oral hygiene will suffice; however, if there is evidence of systemic upset (lymphadenopathy) metronidazole 200 mg tds for 3 days is indicated. It is wise to get the patient to rinse with chlorhexidine gluconate 0.2% prior to ultrasonic scaling to reduce aerosol spread. Chlorhexidine rinses may also be prescribed as an adjunct to brushing, which is painful initially. Later Rx, such as gingivectomy for persistent craters, is only rarely required.
Periodontal abscess is a localized collection of pus within a periodontal pocket. It occurs either due to the introduction of virulent organisms into an existing pocket or ↓ drainage potential. The latter classically occurs during Rx as reduction of inflammation in the coronal gingival tissues occludes drainage by a tighter adaptation to the tooth. May also occur due to impaction of a foreign body such as a fishbone in a pre-existing pocket or even in an otherwise healthy periodontal membrane.
Diagnosis. Need to distinguish from apical abscess.
| Apical abscess | Periodontal abscess |
| Non-vital | Usually vital |
| TTP vertically | Pain on lateral movements |
| May be mobile | Usually |
Insertion of a GP point into an associated sinus and a radiograph may be helpful.
Rx. Emergency : Incision and drainage under LA; debridement of the pocket (e.g. ultrasonic scaler); systemic antibiotic, e.g. metronidazole 200-400 mg tds and/or amoxicillin 250-500 mg tds for 5 days if systemic involvement. Follow-up : conventional Rx for periodontal pockets, combined periodontal-endodontic lesion.
Acute herpetic gingivostomatitis is not described here.
Acute streptococcal gingivitis. Rare. Beefy, red, painful gingivae usually caused by a Lancefield A streptococcus. Rx: penicillin V 500 mg qds for 7 days and OHI.
PERIODONTITIS IN CHILDREN
There are three main variants: localized aggressive periodontitis, generalized aggressive periodontitis, and prepubertal periodontitis. Originally termed 'periodontosis' (1942), they have become better defined in recent years.
Localized aggressive periodontitis (LAP)
Occurs in children and adolescents. Usually localized to upper and lower incisors and first molars. Retrospective studies have suggested it may affect primary dentition. The gingivae around affected teeth may appear entirely normal despite deep periodontal pockets. The degree of periodontal destruction seems out of proportion to the deposits of plaque and calculus.
Prevalence is low (~0.2%). Afro-Caribbeans (2.5%) > Asians > Caucasians. F > M and often familial.
Microbiology. A range of rare organisms have been cultured. Actinobacillus actinomycetemcomitans (Aa) is the major culprit. Others which may act in association include Eubacterium- and Bacteroides- like spp. Aa has been implicated because of high levels in LAP pockets, I serum antibodies to Aa in LAP patients, and the ability of Aa to produce a range of periodontopathic mediators such as leucotoxin, chemotactic inhibition factor, fibroblast growth inhibitor, and an endotoxic cell wall. These patients do show functional defects of neutrophils. There is some evidence that Aa invades local host tissue.
Rx. Meticulous oral hygiene, conventional scaling, and root planing, often in conjunction with access flap surgery. Consider systemic tetracycline (oxytetracycline 250 mg qds or doxycycline 100 mg od for 2-3 weeks) or locally deposited (e.g. slow release) tetracycline. 3-monthly monitoring of subgingival microflora for Aa with targeted Rx is ideal.
Generalized aggressive periodontitis (GA)
A disease of older children; often beginning around puberty. Unlike LAP, these patients have marked periodontal inflammation, and heavy plaque and calculus deposits.
Prevalence. Figures from the USA suggest ~0.13% in adolescents.
Microbiology. High levels of non-motile, Gram negative facultative anaerobes, including Porphyromonas gingivalis. In common with LAP these patients have neutrophils with ↑ chemotaxis.
Rx. As above optimal antibiotic not yet known.
Prepubertal periodontitis
Rare. Appears soon after eruption of primary dentition. Characterized by severe gingival inflammation, and rapid bone and tooth loss.
Microbiology. Usual periodontal pathogens.
Pathology. Thought to be due to a defective leucocyte glycoprotein, preventing leucocyte migration and causing ↑ effective immune response.
Exclude hypophosphatasia before making diagnosis, as cementopathia seen in this can mimic prepubertal periodontitis.
Rx. As for LAP but avoid tetracycline. Use amoxicillin 125-250 mg tds instead.
Chediak-Higashi is not described here.
Papillon-Lefevre is not described here.
PREVENTION OF PERIODONTAL DISEASE
Overview
Following the comments on the pages covering the aetiology and epidemiology of periodontal disease, it is quite clear that dental plaque is the cause of the problem and its elimination will prevent gum disease. This is easier said than done -- remember most of the world's population has gingivitis and/or periodontitis. The key to prevention is regular and thorough plaque removal, therefore OHI is probably the most useful advice you can give to your patients. Smoking exacerbates periodontal disease and adversely affects Rx outcome. Patients should be advised of this.
OHI should include an explanation of the nature of the patient's disease and hence the reasons for good oral hygiene. Identify and demonstrate to the patient the disease (swollen gingivae, bleeding on probing) using a hand mirror, then demonstrate the cause (plaque), either directly, by scraping off a deposit, or by disclosing. Explain how plaque starts to grow immediately after toothbrushing, so that regular removal is necessary, and that it cannot be rinsed away. Then demonstrate how to remove it, avoiding overt criticism of the patient's present efforts, as this is often counterproductive.
Toothbrushing requires a brush, ideally with a small head and even nylon bristles (3-4 tufts across by 10-12 lengthways), which should be renewed at least monthly. Toothpaste makes the process more pleasant and is a useful medium for topical fluoride and other agents. Anticalculus pastes ↑ plaque formation by about 50%. Chlorhexidine-containing toothpastes are active against plaque microorganisms. Numerous methods can be described, based on the movement of the brush stroke: rolling, vibratory, circular, vertical, horizontal. The best is the one which works for that patient (as demonstrated by absence of plaque on disclosing after brushing) and does no harm to tooth or gingivae. The horizontal scrub is notorious for possibly exacerbating gingival recession. Modifications to toothbrushes and brushing technique are often required in children, the elderly, and those with disabling diseases.
Interdental cleaning. Brushing alone is unlikely to clean the interdental spaces adequately; however, it must be mastered first by the patient, before interdental cleaning is taught. Flossing, mini interdental brushes (particularly good for concave root surfaces), and interspace brushes are available for cleaning interproximally. The use of dental floss is something of an art form and must be learned by demonstration.
Professional preventive techniques
Regular periodic examination for periodontal disease, which is largely asymptomatic, is essential. This requires a full oral examination, including probing for pockets as part of routine examination (look above). In patients who have been demonstrated to have periodontal disease, 3-monthly follow-ups are advisable. Routine scaling and polishing is of little value unless accompanied by intensive education and motivation of the patient (however tedious this may be), as it is the patient's efforts which will prevent the re-formation of plaque and initiation of periodontal disease. Employment of a hygienist has obvious benefits in this area. Elimination, avoidance, and Rx of iatrogenic problems such as overhanging margins on restorations, ill-fitting crown margins, poorly designed appliances, etc., are mandatory.
PRINCIPLES OF TREATMENT
- Establish diagnosis.
-
Periodontal disease is an infection due to the presence of plaque, therefore control of plaque is the key to success. More complex Rxs will always fail in the absence of effective plaque control.
- The aims of corrective techniques such as scaling, root planing, periodontal surgery, restorative
work, endodontics, and occlusal adjustment, etc. are:
-
to eliminate pathological periodontal pockets, or to create a tight epithelial attachment where the
pocket once existed; - to arrest loss of, and in some cases improve, the alveolar bone support;
- to create an oral environment which is relatively simple for the patient to keep plaque-free.
The overall aim could be summarized as the creation of a healthy mouth which the patient is both capable of, and willing to, maintain.
It is often convenient to divide the principles of periodontal therapy into three phases:
-
The initial (cause-related) phase, where the aim is to control or eliminate gingivitis and arrest any further progression of periodontal disease by the removal of plaque and other contributory factors.
- The corrective phase is designed principally to restore function and, where relevant, aesthetics.
-
The maintenance (supportive) phase aims to reinforce patient motivation so that their OH is adequate to prevent recurrence of disease. This phase is receiving ↑ attention due to the relative ease with which disease activity can be monitored by probing and chairside diagnostic arrays.
NON-SURGICAL THERAPY-1
Overview
Non-surgical therapy in periodontal disease consists o f debridement, restorative Rx (to correct coexisting or exacerbating factors, e.g. periapical infection, overhanging margins), and the use of antiseptics and antibiotics.
Scaling is the removal of plaque and calculus from the tooth surface, either with hand instruments (e.g. curettes, hoes, chisels, and jaquettes) or mechanically (e.g. Cavitron). Scaling can be sub- or supragingival depending upon the site of the deposits. LA is usually not necessary. It is customary to make use of an ultrasonic scaler for the bulk of the work and finish off, particularly subgingivally, with hand instruments. The precise use of hand instruments is largely a matter of personal preference; however, it is essential to use controlled force and a secure finger-rest. Ultrasonic instruments are quicker, but they can be uncomfortable and leave an uneven root surface (though the significance of the latter is controversial). Ultrasonic scaling employs a frequency of 25-40 000 cycles/sec. Another instrument, known as a sonic scaler and vibrating at 1600-1800 cycles/sec, has been shown to be equally effective at removing calculus and may result in smoother root surfaces. With both it is essential to use a copious coolant spray.
The teeth are usually polished after scaling, preferably using a rubber cup and a fluoride-containing paste (e.g. toothpaste). Patients can then appreciate the feeling of a clean mouth, which they must then maintain.
Local delivery of medicaments
Due to controversy over the efficacy and unwanted effects of systemic antibiotics, methods of direct delivery into the pocket have been explored. These have included using injected pastes or gels, or by impregnated fibre. This gives a high local dose, low systemic uptake, and prolonged exposure of the pathogens to the drug. The rate of crevicular fluid turnover is such that the substansivity of these agents is low. Examples: Blackwell Dentomycin (minocycline), Colgate-Palmolive Elyzol (metronidazole), and Periochip (chlorhexidine). However, only small ↑ in attachment levels have been reported and currently local adjunctive antibiotics are only indicated for the Rx of persistent local defects.
Antimetabolites
NSAIDs are the main group being investigated. Experimental gingivitis and alveolar bone loss in animals are ↑ by flurbiprofen. This is not only due to cyclo-oxygenase pathway inhibition. Evidence of significant long-term benefit in humans is awaited. A future possibility is inclusion of NSAIDs in topical form in a toothpaste.
NON-SURGICAL THERAPY-2
Antiseptics and antibiotics
As the mechanical control of plaque is a tedious and time-consuming procedure, the use of chemicals either as antiseptics or antibiotics would be a significant aid. Problems with their use include the unwanted side-effects of such chemicals, development of resistance, mode of delivery, and most importantly, the fact that no single organism has been identified as the main pathogen in periodontal disease. Nevertheless, elimination of Gram negative anaerobic bacteria has been demonstrated to result in improved periodontal health.
Antiseptics. The antiseptic of greatest proven value is chlorhexidine gluconate. This is commonly used in a 0.2% mouthwash or gel, although 0.12% mouthwash is also available. A standard regimen is 10 ml of solution rinsed for 1 min bd. The gel can be used instead of toothpaste. NB An interaction between conventional toothpaste and chlorhexidine reduces the antiseptic's efficacy. Other proprietary antiseptic rinses have yet to meet this 'gold standard'.
Antibiotics. A review of the use of antibiotics in periodontology recommended their use for patients with recent, or a high risk of, periodontal breakdown. Antibiotics are generally used as an adjunct to mechanical non-surgical therapy in early onset periodontitis and some forms of adult periodontitis. Short-term, relatively high doses, or high locally delivered doses, are used. The two most useful drugs are:
Tetracycline. Can be administered either systemically or directly into the pocket via a slow release mechanism (Dentomycin gel). It is active against spirochaetes, Actinobacillus actinomycetemcomitans, and many other periodontal pathogens. Most useful in the Rx of LAP. Oxytetracycline and doxycycline are currently popular. In addition to being antibacterial, tetracyclines ↓ host neutrophil collagenases and ↓ bone loss. Crevicular fluid concentrations are high. Currently there is much interest in doxycycline (20 mg bd for 3/12). At this dose it has no detrimental effect on the periodontal microflora and its action is mainly to reduce collagenolytic metalloproteinases.
Metronidazole. Effective against protozoa and strict anaerobes and capable of eliminating all the strict anaerobes found in periodontal pockets. As it does not interfere with aerobes or facultative anaerobes it is highly unlikely to allow the development of opportunist pathogens. There is frequent mention of the mutagenicity and teratogenicity of this drug in animal studies but, despite widespread use in medicine, no clinical evidence has emerged to support this. It is, in fact, an extremely safe and useful drug. When combined with mechanical pocket therapy, significant improvements in terms of probing depth have been demonstrated. There is an emetic interaction with alcohol.
There is a clear place for antibiotics/antiseptics as adjuncts in selected types of periodontal disease, e. g. aggressive periodontitis and acute or severe periodontal infections.
MINIMALLY INVASIVE THERAPY-1
Overview
There has been a progressive move away from pocket elimination surgery, towards the creation of a healthy periodontium which gives access for both professional and home cleaning. Recognition that periodontal disease is a localized infection due to the presence of dental plaque, and that its arrest and prevention is dependent on the removal of plaque and plaque-retaining factors, has allowed a far more rational and conservative approach to therapy to develop. The prime aims of periodontal surgery are now to:
- remove deposits from root surfaces;
-
create subgingival root surfaces which are accessible for cleaning, either by professionals or by the patient;
- maximize the potential for healing of damaged periodontal tissues.
Minimally invasive techniques include deep subgingival scaling/root planing and the modified Widman flap (open flap debridement). These are suitable for those patients who, despite good supragingival plaque control, still have true pockets but do not need recontouring of the gingival margins.
Scaling and root planing. Deep subgingival scaling and root planing are best carried out under LA. Deep scaling is a procedure which removes plaque and calculus from the root surface; it differs from supragingival scaling simply in its thoroughness, and the discomfort it causes. Root planing is a technique whereby endotoxin-damaged cementum is removed from the root surface by scraping the root. The same instruments are used for both procedures, and in practice it is probably impossible to differentiate between subgingival scaling and root planing, as in both techniques plaque, calculus, root cementum, and small amounts of dentine will be removed. These are the core procedures for all periodontal techniques; the only real difference with formal surgery is that it allows Rx under direct vision, osseous recontouring and sculpting if desired, and repositioning of the gingiva.
It is often most effective to treat one quadrant at a time under LA, partly because it is painful and partly because it is tedious when performed meticulously, which is the only worthwhile way to do it. It has been sugested, however, that full mouth root planning be done in two visits separated by only 24 h, with concurrent use of a chlorhexidine mouthwash. The choice of instruments is less important than the end result, as effective debridement of the root surface is essential. It is not necessary to curette the epithelial lining of the pocket. Success will allow tight adaptation of the pocket epithelium to the root, creating a long junctional epithelium.
As smoking ↓ outcome of Rx, some periodontologists have limited Rx in those continuing to smoke. While periodontal surgery hardly has the public impact of cardiac surgery, the ethical problem is the same.
Practical tips for periodontal surgery
Local anaesthesia. The infiltration, block and/or lingual/palatal injections required will be determined by the site of surgery. Both LA and haemostasis are improved by injecting directly into the gingival margin and interdental papillae until blanching is seen.
Suturing techniques. Interrupted interproximal sutures are used when buccal and lingual flaps are being reapposed at the same level. When flaps are repositioned at different levels, a suspensory suture is used, where the suture only passes through the buccal flap and is suspended around the cervical margins of the teeth.
Periodontal packs. These are essential after gingivectomy to ? post-operative discomfort. Many favour them after all periodontal surgery to help reappose the flap to bone.
Classified:
- Eugenol dressings, e. g. ZnO, have the advantage of being mildly analgesic but can cause sensitivity reactions.
- Eugenol-free dressings, e.g. Coe-pack, are more popular.
MINIMALLY INVASIVE THERAPY-2
The modified Widman flap
This is a technique which enables open debridement of the root surface, with a minimal amount of trauma. There is no attempt to excise the pocket, although a superficial collar of tissue is removed. This has the advantage of allowing close adaptation of the soft tissues to the root surface with minimal trauma to, and exposure of, underlying bone and connective tissue, thus causing fewer problems with post-operative sensitivity and aesthetics.
Technique (after Ramfjord and Nissle 1974). A scalloped incision is made parallel to the long axis
of the teeth involved 1 mm from the crevicular margin, except when pockets are >2 mm deep when
an intracrevicular incision is made. This incision is extended interproximally as far as possible,
separating the pocket epithelium from the flap to be raised, and then extended mesially and
distally, allowing the flap to be raised as an envelope without relieving incisions. The flap should be
as conservative as possible and only a few millimetres of alveolar bone exposed by a second
incision intercrevicularly to release the collar of pocket epithelium and granulation tissue. A third
incision at 90 degrees to the tooth separates the pocket epithelium, and this is removed along with
accompanying granulation tissue with curettes and hoes. The root surface is then thoroughly scaled
and root planed. Although bony defects can be curetted no osseous surgery is carried out. The flaps
are then repositioned to cover all exposed alveolar bone and sutured into position. Post-operatively
chlorhexidine 0.2% 10 ml rinsed bd is given, and most periodontologists prefer to use a periodontal
pack to improve patient comfort post-operatively.
Notes. There continues to be considerable confusion about nomenclature with this type of flap. The original Widman flap described in 1918 by Leonard Widman used relieving incisions and osseous surgery, and attempted to remove the pocket. The Neumann flap used an intracrevicular incision, again attempting to excise the pocket, but the 'modified flap operation' described by Kirkland in 1931 did not require sacrifice of inflamed tissues or apical displacement of the gingival margin. The latter was, of course, the precursor of the 'modified Widman flap'.
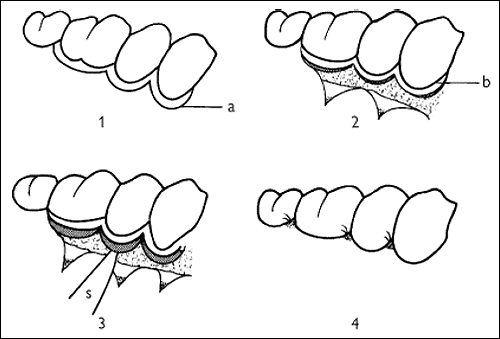
Modified Widman flap
- Design of flap
a Incision - Flap elevated
b Gingival cuff to be discarded - Excision of supra-alveolar pocket
s Scalpel blade - Flap repositioned and sutured in place
PERIODONTAL SURGERY
Gingivectomy
Has ↓ in use over the last decade; its remaining indications are in cases with persistence of deep supra-alveolar pockets (e. g. gingival hypertrophy), to reshape severely damaged gingivae into an easily manageable contour to treat gingival overgrowth, and for crown lengthening prior to restorative procedures. It is not suitable for the management of deep 'true' pocketing as excision of the pocket will remove the entire thickness of keratinized gingivae. It is of no value in the Rx of infrabony lesions.
Technique. Pockets are delineated by use of pocket marking forceps, e.g. Crane-Kaplan forceps. This marks out a line of incision, which may be either smooth or scalloped, made with the blade angled at 100-110° to the long axis of the tooth. This bevelled incision excises supragingival pockets and allows for gingival recontouring. Once the incision has been made the strip of gingiva remaining is released by an intercrevicular incision. The root surfaces are then curetted and an open area of freshly cut granulation tissue left to heal under a periodontal pack. Prescribe chlorhexidine mouthwash 10 ml bd. The pack is left in place for about 1 week.
Disadvantages. Loss of attached gingiva, raw wound, exposed root surface (which ↑ likelihood of sensitivity and caries). Some remodelling of alveolar bone occurs, despite there being no operative interference.
Apically repositioned flap
This procedure is used to expose alveolar bone and includes the option for osseous surgery to correct infrabony defects. It allows excellent access to the root surface for debridement. The principal difference between this procedure and the modified Widman flap is the deliberate exposure of alveolar bone, and the apical repositioning of the flap with post-operative exposure of the root surfaces. This is primarily a buccal procedure, and although it can be performed on lingual pockets, it is obviously impossible on the palate where a conventional or reverse bevel gingivectomy approach has to be used.
Technique. A reverse bevel incision is made in the attached gingiva angled to excise the periodontal pocket in a scalloped outline with vertical relieving incisions at either end. A split thickness flap is made down to bone and then converted to full thickness, leaving a residual collar of tissue around the root surfaces. This combination of pocket epithelium and granulation tissue is removed with a curette. If indicated the alveolar crest can be remodelled.
Advantages. Include exposure of alveolar bone with controlled bone loss, exposure of furcation area, minimal post-operative pocket depth, ability to reposition the flap, and primary closure of the wound. In addition, keratinized gingiva is preserved.
Disadvantages. Exposure of root surface (leading to ↑ susceptibility to caries and sensitivity) and ↑ loss of alveolar bone height, which accompanies full exposure of the bone at operation.
Osseous surgery
Bone recontouring has become less popular as it is always accompanied by some degree of alveolar resorption and therefore ↓ support for the tooth. Osteoplasty is conservative recontouring of the bone margin (i. e. non-supporting bone).
Ostectomy is excision of bone aimed at eliminating infra-alveolar pocketing, but unfortunately it also ↓ alveolar support. The aim of osseous surgery should be to establish a more anatomically correct relationship between bone and tooth while maintaining as much alveolar support as possible.
Other flap procedures
These include simple replaced flaps which give ↑ bony access compared to the modified Widman
flap. Also crown-lengthening procedures, which can range from a simple gingivectomy to an apically
repositioned flap ± bone removal. In addition, many periodontologists have their own modification
of the aforementioned techniques.
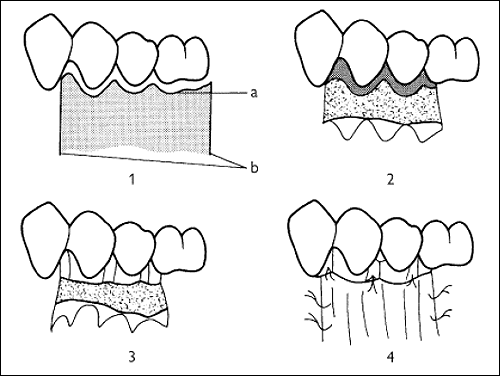
Apically repositioned flap
- Design of flap
(a) Reverse bevel incision
(b) Relieving incisions - Elevating the flap. Tissue enclosing pocketing which is to be discarded is hatched.
- Flap elevated, pockets excised. Osseous surgery can be performed at this stage.
- Flap apically repositioned and sutured in position.
REGENERATIVE TECHNIQUES
Guided-tissue regeneration
The recognition that epithelium migrated along the root surface before any other cell type, after
periodontal surgery, and created the long junctional epithelium which prevented new attachment,
created the possibility that prevention of migration of epithelium would allow new connective tissue
attachment. Guided-tissue regeneration (GTR) is essentially interposing a barrier to epithelial
migration prior to completion of surgical or non-surgical therapy. Original barriers were Millipore
and PTFE. Goretex membranes in a variety of shapes are now widely used and infrabony and
furcation defects can be infilled using a combination of bone/hydroxyapatite/biocoral covered by the
membrane. Goretex has to be removed at 4-6 weeks. Biocompatible resorbable materials, such as
Vicryl and lyophilized collagen (Resolute) allow a one-operation technique. Tissue growth factor β-2
and bone morphogenetic protein may have a role in stimulating bone and connective tissue
deposition. Main disadvantage is cost. Please note that a recent systematic review has also
questioned the effectiveness of these techniques.
Endogain is a product containing enamel matrix derivative (EMD) proteins. These substances (e. g. amelogenin) are found in Hertwig's sheath and induce root formation in the developing tooth. Locally applied enamel matrix proteins may help form acellular cementum, the key tissue in the development of a functional periodontium.
Technique. Access to the root surface is gained surgically, the cementum is mechanically cleaned, and EMD solution is applied to the root surfaces. The access flaps are then repositioned and sutured.
Outcome. Regeneration of cementum, periodontal ligament, and alveolar bone appears to be
possible experimentally; long-term clinical outcome awaits prospective RCCTs.
PERI-IMPLANTITIS
Osseo-integrated implants are now well established. Implants which have achieved full osseo-integration may fail by overloading, peri-implantitis (similar to periodontitis), or a combination of both, with failure rates being high in smokers. Implant salvage in the failing stage consists of ↓ overloading and the entire arsenal of periodontal therapies. Local antibiotics and bonesupplemented GTR may be particularly useful.
Tissue transformation using bone morphogenetic protein may also prove useful in the future.
MUCOGINGIVAL SURGERY
Overview
Mucogingival surgery encompasses those techniques aimed at the correction of local gingival defects. The rationale for this type of surgery has been hotly debated over many years. Initially, it was felt that a margin of attached gingiva of around 3 mm was required to protect the periodontium during mastication and to dissipate the pull to the gingival margin from fraenal attachments. In fact, data from properly conducted experimental work have demonstrated that the width of attached gingiva and the presence or absence of an attached portion are not of decisive importance for the maintenance of gingival health. As a result of this, the indications for mucogingival surgery have been rationalized:
-
where change in the morphology of the gingival margin would improve plaque control, e.g. presence of high fraenal attachments or deep areas of recession;
- areas where recession creates root sensitivity or aesthetic problems;
-
a very thin layer of attached gingiva overlying a tooth which is to be moved orthodontically: the
evidence for this is somewhat anecdotal.
Gingival recession
Gingival recession is one of the commonest reasons for carrying out mucogingival surgery. The two commonest causes are plaque-induced gingival inflammation and toothbrush trauma, revealing dehiscences in alveolar bone. Therefore, basic periodontal care and correction of faulty toothbrushing technique are the first lines of Rx. While anatomical features may contribute, these and trauma from occlusion, high fraenal attachments, and impingement from restorations, etc., are a secondary consideration.
Mucogingival techniques
These can be divided into two main groups:
Vestibular extension procedures essentially aim to ↑ the area of attached gingivae and deepen the
buccal or labial sulcus. As they are always accompanied by a degree of bone resorption, these
methods are not recommended as a therapeutic periodontal technique.
Grafting is subdivided into:
-
Free grafts, which are completely removed from their donor area. Free gingival grafts, commonly of palatal mucosa and connective tissue, are taken and grafted to donor sites prepared by incising between attached and alveolar mucosa. While this technique may successfully cover exposed root surfaces of around 2 mm square and will certainly ↑ the width of keratinized gingiva, long-term cross-over studies suggest that in the presence of meticulous OH there is no significant difference between attachment levels in grafted and non-grafted sites with similar degrees of recession.
-
Pedicle grafts are not separated from their blood supply. Commonly used pedicle grafts are the laterally repositioned flap, coronally repositioned flap, and the double papilla flap. These techniques may be of some value in very narrow areas of isolated gingival recession. Technically, of course, these are flaps not grafts.
REATTACHMENT/NEW ATTACHMENT
Definitions
Reattachment is defined as the reunion of connective tissue and root separated by incision or injury. New attachment is defined as the reunion of connective tissue with a root surface which has been pathologically exposed (i.e. due to periodontal disease). It is the ideal aim of periodontal therapy.
There are two ways in which new attachment may occur; these can be subdivided anatomically as
those occurring within bony pockets and those occurring between the soft tissue of the previously
existing periodontal pocket and the root surface. There have been numerous claims and
counterclaims as to which of these may occur, following virtually all forms of periodontal
intervention. However, there is very little satisfactory evidence to support the contention that new
attachment takes place above the level of the alveolar crest. More recently, re-entry procedures in
animals have suggested that new attachment formation is inhibited by the apical migration of
dento-gingival epithelium forming a long, but apparently quite healthy, long junctional epithelium.
Some newer procedures now describe methods to inhibit apical migration of dento-gingival junction
epithelial cells in an attempt to obtain new attachment. However, animal experiments suggested
that granulation tissue originating from bone or gingival connective tissue was unable to establish
new connective tissue attachment, even when the intervening dento-gingival epithelium was
prevented from migrating into the treated area. In fact, when close apposition of connective tissue
to root surface occurred, resorption and ankylosis often resulted, suggesting that the migration of
dento-gingival epithelium into the Rx area may be a protective mechanism. Experiments to assess
whether this behaviour also applied to the PDL have been carried out. Interestingly, it would
appear that PDL cells may develop new attachment if dento-gingival junction epithelial cells are
prevented from migrating into the area, without the occurrence of resorption and ankylosis. This
has been confirmed in clinical studies, using a variety of materials to form a barrier against ingress
of epithelial cells by a process known as GTR). This involves placement of a mechanical
barrier (Goretex, Vicryl, Resolute) underneath the flap, extending from the outer surface of the
alveolar process to the crown of the tooth above the gingival margin. This allows preferential
colonization by PDL cells. Resorbable barriers avoid a retrieval operation.
EMD is not described here.
Bony infill in osseous defects. There are a number of studies which suggest that complete
regeneration can occur in up to 70% of three-walled infrabony defects. This success rate, however,
is not consistent, especially in combined and two-walled defects.
OCCLUSION AND SPLINTING
All matters relating to occlusion seem to have developed a high degree of mysticism about them in
the dental world; this is also true of the relationship between occlusion and periodontal therapy.
It used to be claimed that angular bony defects and ↑ mobility were directly attributable to trauma
from the occlusion. This belief is less commonly held nowadays as angular defects can be found
around both occlusally traumatized and non-traumatized teeth. It is, however, self-evident that
an already periodontally diseased tooth can change its relationship in the arch to become
traumatized, or that a tooth which is already in traumatic occlusion can develop periodontal
disease, and that the two factors can exacerbate one another.
Tooth mobility. ↑ tooth mobility may simply be a result of loss of periodontal attachment and bony
support. It may also result purely as a localized effect due to a heavy occlusal loading, causing a
widening of the periodontal membrane space, though this is usually iatrogenic in origin. It is now
felt that the diagnosis of occlusal trauma should only be made where progressive increasing tooth
mobility is observed, but in order to do this it is necessary to have an objective method of
measuring tooth mobility. This can be done using a Mobility Index:
Grade 1 = Mobility <1 mm buccolingually.
Grade 2 = Mobility 1-2 mm buccolingually.
Grade 3 = Mobility of >2 mm buccolingually and/or vertical mobility.
Rx. First priority should be to diagnose and treat any existing periodontal disease and correct any
pre-existing iatrogenic causes, e.g. poor crowns or bridges, high restorations. If tooth mobility
persists as a direct result of diagnosable occlusal trauma, occlusal adjustment is a sensible Rx
modality. If the tooth is mobile as a result of lack of alveolar bone support, this is not automatically
an indication for splinting (see below).
Splinting. Indicated in the following situations:
- Tooth with healthy but ↓ periodontium where mobility is ↑.
- Tooth with ↑ mobility which patient finds uncomfortable during function.
It is very easy to design splints which are impossible for the patients to keep clean as all additions
to the natural tooth surface will ↑ plaque retention. A wide range of different techniques and
materials have been described, including orthodontic wire fastened to teeth by resin-composite,
resin-composite alone, fixed bridges, partial prostheses, acid-etch retained splints, and, more
recently, fibre-reinforced resin-composite splinting.
PERIO-ENDO LESIONS
Overview
! It is essential to vitality test any heavily restored tooth with periodontal involvement.
Given the relative frequency of both periodontal disease and periapical pathology, it is not
surprising that both may occur together, which can result in diagnostic confusion. In fact, there is
little evidence to support the popular notion that periodontitis leads to pulp necrosis. However,
there is no doubt that pulp pathology can exacerbate periodontal problems.
Pulpal problems
Acute pulpitis is not described here.
Non-vital pulp may cause asymptomatic periapical lesion or periapical abscess.
Lateral canal and/or non-vital pulp may mimic periodontal abscess, as can a root perforation following endodontic therapy.
Vertical root fracture and/or non-vital pulp can lead to periodontal inflammation and may mimic periodontal abscess.
Horizontal root fracture may mimic periodontal abscess.
Periodontal pathology and its effect on the pulp
Deep pocketing may encroach on lateral canals in the apical 1/3 of the root, but is otherwise unlikely to cause direct pulpal pathology.
Gingival recession is directly associated with hypersensitivity of root dentine.
Root planing and furcation procedures actively involve dentine, and can clearly lead to hypersensitivity and sometimes acute pulpal changes.
Differential Diagnosis
Primarily periodontal | Primarily pulpal | |
| History | No preceding toothache | Often toothache |
| Percussion | TTP especially lateral | TTP especially vertical |
| Probing | Pocketing always | May be no pockets |
| Probing sinus | May lead to pocket | May lead to apex |
| Vitality test | Usually positive | Negative |
| Radiographs | Vertical bone loss | Apical area |
Combined perio-endo lesions
These may be either:
-
Coexisting, but separate from each other, in which case standard endodontic and periodontal therapy are used as indicated.
-
Interconnected, in which case probing both pocket and sinus will reach the apex. This can be confirmed by taking a periapical film with a GP point inserted into the pocket.
Rx of combined interconnected lesion. First, resolve the acute infection and inflammation by drainage (and/or antibiotics), then treat with orthograde RCT (the greater the pulpal component the better the prognosis).The apparent periodontal lesion will often be seen to resolve to a substantial degree over a period of months,therefore the decision to carry out surgery should be deferred. Combined apicectomy and periodontal surgery is quite feasible but carries a poorer long-term prognosis. The worst prognosis applies to those teeth where the periapical/pulpal pathology has been due entirely to apical extension of the periodontal pocket. These are often diagnosed after the fact, when endodontics completely fails to resolve the lesion.
FURCATION INVOLVEMENT
Overview
The extension of periodontal disease into the bi- or trifurcation of multirooted teeth is known as furcation involvement.
Diagnosis is established by probing into the furcation and by radiographs. The possibility of pulpal pathology is ↑ in teeth with furcation involvement and vitality testing is essential. Radiographs give a guide to the degree of alveolar bone loss both mesially and distally, and in the furcation area.
Classification
1st degree: horizontal loss of support not exceeding 1/3 tooth width. Requires scaling and root planing, possibly with furcation plasty.
2nd degree: horizontal loss of support exceeding 1/3 but not encompassing the total width of the furcation area. May require furcation plasty, and/or tunnel preparation, and/or root resection, and/or extraction.
3rd degree: horizontal through-and-through destruction in the furcation area. May require tunnel preparation, and/or root resection, and/or extraction.
Rx techniques
Scaling and root planing. Unless the post-Rx morphology can be kept clean by the patient it will not be successful.
Furcation plasty. An open procedure involving a muco-periosteal flap to allow root planing and scaling, followed by the removal of tooth structure in the furcation area to achieve a widened entrance to give access for cleaning. Osseous recontouring may be used if indicated. The flap is repositioned and sutured to ↑ access post-operatively. There is an obvious risk of pulpal damage and post-operative dentine sensitivity.
Tunnel preparation is a similar procedure to furcation plasty using buccal and lingual flaps, the main
difference being that the entire furcation area is exposed and the flaps are sutured together intraradicularly
to leave a large exposed furcation. There is a high risk of post-operative caries, dentine
sensitivity, and pulpal exposure, making this a method to be used with caution. It is of most value
for mandibular molars in patients with optimal OH. In many cases considered for furcation plasty or
tunnelling, it may be more sensible to proceed to a more radical approach such as root resection.
Root resection. Involves amputation of one (or even two) of the roots of a multirooted tooth, leaving the crown and the root stump. It is important to ensure that the root to be retained can be treated endodontically, is in sound periodontal state with good bony support, is restorable, and will be a viable tooth in the long term. At operation it is wise to raise a flap to enable direct visualization of the root surface. Resection of the root with a high-speed bur is followed by smoothing, recontouring, and restoration of any residual pulp cavity. It is sometimes not possible to proceed with root resection, despite apparently favourable radiographs, especially in maxillary molars, so warn patient pre-operatively.
Hemisection. Involves dividing a two-rooted tooth in half to give two smaller units each with a single root. Again, RCT is necessary pre-operatively and restoration of the divided crown is required postoperatively.
Extraction. Ensures removal of periodontal disease but carries its own problems.
Guided tissue regeneration is not described here.
Enamel matrix derivatives EMD is not described here.
It is important to note that the techniques described above are of less significance to long-term outcome than the degree of plaque control that can be achieved and maintained by the patient. Mini interproximal brushes are a valuable aid in cleaning furcation defects and are available in a variety of sizes and shapes.
