disclaimer &
медицина
урология
painful bladder...
аватары
обо мне
визитки &
css &
клипарт
лит-ра
lola rennt
ссылки &
отметиться
дать денег
гостевая книга
INTERSTITIAL CYSTITIS
AND RELATED DISORDERS
Painful bladder syndrome/interstitial cystitis (PBS/IC) is a condition diagnosed on a clinical basis and requiring a high index of suspicion by the clinician. Simply put, it should be considered in the differential diagnosis of the patient who presents with chronic pelvic pain that is often exacerbated by bladder filling and associated with urinary frequency. The perception that the original term, interstitial cystitis, was not at all descriptive of the clinical syndrome or even the pathologic findings in many cases has led to the current effort to reconsider the name of the disorder and even the way it is positioned in the medical spectrum. What was originally considered a bladder disease is now considered a chronic pain syndrome that may begin as a pathologic process in the bladder in most but not all patients and eventually can develop into a disease in a small subset of those affected that even cystectomy may not benefit. Its relationship to other chronic pelvic pain syndromes including the chronic pelvic pain syndrome in men (previously referred to as nonbacterial prostatitis) is unclear.
PBS/IC encompasses a major portion of the "painful bladder" disease complex, which includes a large group of patients with bladder and/or urethral and/or pelvic pain, irritative voiding symptoms (urgency, frequency, nocturia, dysuria), and sterile urine cultures. Painful bladder conditions with well-established causes include radiation cystitis, cystitis caused by microorganisms that are not detected by routine culture methodologies, and systemic disorders that affect the bladder. In addition, many gynecologic disorders can mimic PBS/IC.
The symptoms are mostly allodynic, an exaggeration of normal sensations. Urinary frequency patterns can be related to fluid intake and age, and the signal or urge to void is considered an unpleasant or painful sensation by most persons. There are no pathognomonic findings on pathologic examination, and even the finding of petechial hemorrhages on the bladder mucosa during cystoscopy after bladder hydrodistention under anesthesia is no longer considered the sine qua non of PBS/IC that it had been until a decade ago. PBS/IC is truly a diagnosis of exclusion. It may have multiple causes and represent a final common reaction of the bladder to different types of insult.
HISTORICAL PERSPECTIVE
Recent historical reviews confirm that IC was recognized as a pathologic entity during the 19th century. Joseph Parrish, a Philadelphia surgeon, described three cases of severe lower urinary tract symptoms in the absence of a bladder stone in an 1836 text and termed the disorder "tic doloureux of the bladder". Teichman and colleagues (2000) argue that this may represent the first description of IC. Fifty years later Skene (1887) used the term interstitial cystitis to describe an inflammation that has "destroyed the mucous membrane partly or wholly and extended to the muscular parietes".
Early in the 20th century, at a New England Section meeting of the American Urological Association, Guy Hunner reported on eight women with a history of suprapubic pain, frequency, nocturia, and urgency lasting an average of 17 years. He drew attention to the disease, and the red, bleeding areas he described on the bladder wall came to have the pseudonym "Hunner's ulcer". As Walsh (1978) observed, this has proven to be unfortunate. In the early part of the 20th century, the very best cystoscopes available gave a poorly defined and ill-lit view of the fundus of the bladder. It is not surprising that when Hunner saw red and bleeding areas high on the bladder wall he thought they were ulcers. For the next 60 years, urologists would look for ulcers and fail to make the diagnosis in their absence. The disease was thought to be a focal cystitis rather than a pancystitis.
Hand (1949) authored the first comprehensive review about the disease, reporting 223 cases. In looking back, his paper was truly a seminal one, years ahead of its time. Many of his epidemiologic findings have held up to this day. His description of the clinical findings bears repeating. "I have frequently observed that what appeared to be a normal mucosa before and during the first bladder distention showed typical interstitial cystitis on subsequent distention". He notes, "small, discrete, submucosal hemorrhages, showing variations in form…dot-like bleeding points…little or no restriction to bladder capacity". He portrays three grades of disease, with grade 3 matching the small-capacity, scarred bladder described by Hunner. Sixty-nine percent of patients had grade 1 disease, and only 13% had grade 3 disease.
Walsh (1978) coined the term glomerulations to describe the petechial hemorrhages that Hand had described. But it was not until Messing and Stamey (1978) discussed the "early diagnosis" of IC that attention turned from looking for an ulcer to make the diagnosis to the concepts that (1) symptoms and glomerulations at the time of bladder distention under anesthesia were the disease hallmarks and (2) the diagnosis was primarily one of exclusion.
KEY POINT: HISTORICAL PERSPECTIVE |
| Bourque's Aunt Minnie (she is hard to define, but you know her when you see her) description of IC is over 50 years old and is worth recalling. "We have all met, at one time or another, patients who suffer chronically from their bladder; and we mean the ones who are distressed, not only periodically but constantly, having to urinate often, at all moments of the day and of the night, and suffering pains every time they void. We all know how these miserable patients are unhappy, and how those distressing bladder symptoms get finally to influence their general state of health physically at first, and mentally after a while." |
Although memorable and right on the mark, Bourque's description under "Key Point: Historical Perspectives" and others like it were not suitable for defining this disease in a manner that would help physicians make the diagnosis and design research studies to learn more about the problem. Physician interest and government participation in research were sparked through the efforts of a group of frustrated patients led by Dr. Vicki Ratner, an orthopedic surgery resident in New York City, who founded the first patient advocacy group, the Interstitial Cystitis Association, in the living room of her New York City apartment in 1984. The first step was to develop a working definition of the disease. The modern history of PBS/IC is best viewed through the perspective of definition.
DEFINITION
Interstitial cystitis (IC) is a clinical diagnosis primarily based on symptoms of urgency/frequency and pain in the bladder and/or pelvis. The International Continence Society (ICS) prefers the term painful bladder syndrome (PBS), defined as "the complaint of suprapubic pain related to bladder filling, accompanied by other symptoms such as increased daytime and night-time frequency, in the absence of proven urinary infection or other obvious pathology". The ICS reserves the diagnosis of IC for patients with "typical cystoscopic and histological features", without further specifying these. In the absence of clear criteria for "IC", this chapter will refer to PBS/IC and IC interchangeably, because all but recent literature terms the syndrome "IC". Perhaps more than for most diseases, how we arrived at this point is instructive and critical to an overall understanding of PBS/IC.
"It resembles a constellation of stars; its components are real enough but the pattern is in the eye of the beholder". This evocative description of fibromyalgia could equally apply to PBS/IC. Indeed, it has been argued, not necessarily convincingly, that each medical specialty has at least one somatic syndrome (irritable bowel syndrome, chronic pelvic pain, fibromyalgia, tension headache, noncardiac chest pain, hyperventilation syndrome) that might be better conceptualized as a part of a general functional somatic syndrome than with the symptom-based classification that we have now that may be more a reflection of professional specialization and access to care.
There are data to suggest that true urinary frequency in women can be defined as regularly having to void at intervals of less than 3 hours, and that of women older than age 40 years, 25% have nocturia at least once. Whereas bladder capacity tends to fall in women by the eighth and ninth decades of life, bladder volume at first desire to void tends to rise as women age. Based on a 90th percentile cutoff to determine the ranges of normality, the highest normal frequency varies in the fourth decade range from 6 voids for men to 9 voids for women. Large variation in the degree of bother with varying rates of frequency makes a symptomatic diagnosis of PBS/IC based on an absolute number of voids subject to question, and frequency per volume of intake or even the concept of "perception of frequency" as a problem may be more accurate than an absolute number.
In an effort to define IC so that patients in different geographic areas, under the care of different physicians, could be compared, the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) held a workshop in August 1987 at which consensus criteria were established for the diagnosis of IC. These criteria were not meant to define the disease but rather to ensure that groups of patients included in basic and clinical research studies would be relatively comparable. After pilot studies testing the criteria were carried out, the criteria were revised at another NIDDK workshop a year later. These criteria are presented in Table 1.
| Table 1. National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) Diagnostic Criteria for Interstitial Cystitis. |
To be diagnosed with interstitial cystitis, patients must have either glomerulations on cystoscopic examination or a classic Hunner ulcer, and they must have either pain associated with the bladder or urinary urgency. An examination for glomerulations should be undertaken after distention of the bladder under anesthesia to 80 to 100 cm H2O for 1 to 2 minutes. The bladder may be distended up to two times before evaluation. The glomerulations must be diffuse — present in at least three quadrants of the bladder — and there must be at least 10 glomerulations per quadrant. The glomerulations must not be along the path of the cystoscope (to eliminate artifact from contact instrumentation). The presence of any one of the following excludes a diagnosis of interstitial cystitis:
|
Although meant initially to serve only as a research tool, the NIDDK "research definition" became a de facto definition of this disease, diagnosed by exclusion and colorfully termed a "hole in the air" by Hald. Certain of the exclusion criteria serve mainly to make one wary of a diagnosis of IC but should by no means be used for categorical exclusion of such a diagnosis. However, because of the ambiguity involved, these patients should probably be eliminated from research studies or categorized separately. In particular, exclusion criteria 4, 5, 6, 8, 9, 11, 12, 17, and 18 are only relative. What percentage of patients with idiopathic "sensory urgency" have IC is unclear. The specificity of the finding of bladder glomerulations has come into question. Similarly, the sensitivity of glomerulations is also unknown, but clearly patients with IC symptoms can demonstrate an absence of glomerulations under anesthesia. Bladder ulceration is extremely rare and accounts for less than 5% of patients in my experience and in the experience of others. A California series found 20% of patients to have ulceration. Specific pathologic findings represent a glaring omission from the criteria, because there is a lack of consensus as to which pathologic findings, if any, are required for, or even suggestive of, a tissue diagnosis.
The unexpected use of the NIDDK research criteria by the medical community as a definition of IC led to concerns that many patients suffering from this syndrome might be misdiagnosed. The multicenter Interstitial Cystitis Database (ICDB) study through NIDDK accumulated data on 424 patients with IC, enrolling patients from May 1993 through December 1995. Entry criteria were much more symptom driven than those promulgated for research studies and are noted in Table 2. In an analysis of the defining criteria, it appeared the NIDDK research criteria fulfilled their mission. Fully 90% of expert clinicians agreed that patients diagnosed with IC by those criteria in the ICDB indeed had the disorder. However, 60% of patients deemed to have IC by these experienced clinicians would not have met NIDDK research criteria. Thus, IC remains a clinical syndrome defined by some combination of chronic symptoms of urgency, frequency, and/or pain in the absence of other reasonable causation. Whereas IC symptom and problem indices have been developed and validated, these are not intended to diagnose or define IC but rather to measure the severity of symptomatology and monitor disease progression or regression.
| Table 2. Interstitial Cystitis Database (ICDB) Study Eligibility Criteria |
|
Recent international consultations have essentially agreed that the nomenclature of "interstitial cystitis" be revised to "painful bladder syndrome/interstitial cystitis". This recognizes that it is the symptoms that drive treatment, and the question as to whether IC refers to a distinct subgroup of the painful bladder syndrome is, as yet, unclear. The International Continence Society (ICS) definition of painful bladder syndrome of "suprapubic pain related to bladder filling, accompanied by other symptoms such as increased daytime and nighttime frequency in the absence of infection or other pathology" appears to be a useful one, highlighting, as it does, that pain is the primary component of PBS/IC. For purposes of PBS/IC, the symptom of pain should be broadened to include "pressure" and "discomfort". IC may be a subgroup that encompasses those patients with typical histologic and cystoscopic features, but what these features are is still controversial and somewhat vague.
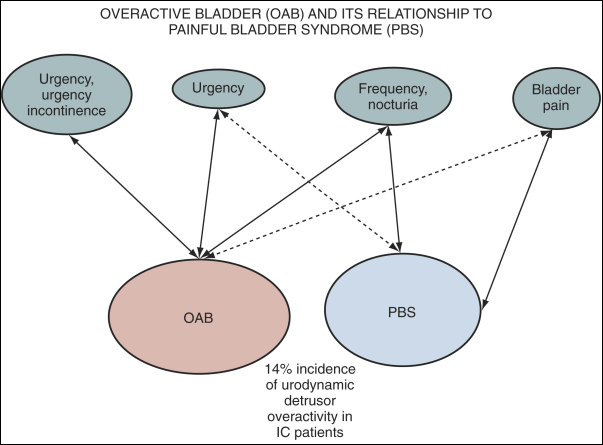
Urgency is a common complaint of this group of patients. The ICS definition of urgency, "the complaint of a sudden compelling desire to pass urine, which is difficult to defer", could be interpreted as compatible with either detrusor overactivity or PBS/IC. Pain and pressure are more involved in the frequency of PBS/IC, and fear of incontinence seems the reason for the urgency of overactive bladder. Thus, urgency is left out of the definition of PBS/IC because it would tend to obfuscate the borders of overactive bladder and PBS/IC and is unnecessary for definition purposes. Figure 1 is a graphic depiction of one view of the relationship between these two, sometimes confused, conditions. The 14% incidence of urodynamic detrusor overactivity in the PBS/IC patients is probably close to what one might expect in the general population if studied urodynamically.
|
| Figure 1. Relationship of overactive bladder (OAB) to painful bladder syndrome (PBS). |
EPIDEMIOLOGY
Epidemiology studies of PBS/IC are hampered by many problems. The lack of an accepted definition, the absence of a validated diagnostic marker, and questions regarding etiology and pathophysiology make much of the literature difficult to interpret. This is most apparent when one looks at the variation in prevalence reports in the United States and around the world. These range from 1.2 per 100,000 population and 4.5 per 100,00 females in Japan to a questionnaire-based study that suggests a figure in American women of 20,000 per 100,00!
It has been estimated that the prevalence of chronic pain due to benign causes in the population is at least 10%. Numerous case series have, until recently, formed the basis of epidemiologic information regarding PBS/IC. Farkas and associates discussed IC in adolescent girls. Hanash and Pool reviewed their experience with IC in men. Geist and Antolak reviewed and added to reports of disease occurring in childhood. A childhood presentation is extremely rare and must be differentiated from the much more common and benign-behaving extraordinary urinary frequency syndrome of childhood, a self-limited condition of unknown etiology. Nevertheless, there is a small cohort of children with chronic symptoms of bladder pain, urinary frequency, and sensory urgency in the absence of infection who have been evaluated with urodynamics, cystoscopy, and bladder distention and have findings consistent with the diagnosis of PBS/IC. In Close and colleague's review of 20 such children, the median age at onset was younger than 5 years and the vast majority of patients had long-term remissions with bladder distention.
A study conducted at the Scripps Research Institute included 374 patients at Scripps as well as some members of the Interstitial Cystitis Association, the large patient support organization. A more recent, but similar study in England concurred with the Scripps findings of urgency, frequency, and pain in the vast majority of these patients, devastating effects on quality of life, and often unsuccessful attempts at therapy with a variety of treatments. Although such reviews provide some information, they would seem to be necessarily biased by virtue of their design.
Several population-based studies have been reported in the literature, and these studies tend to support the reviews of selected patients or from individual clinics and the comprehensive follow-up case-control study by Koziol. The first population-based study included "almost all the patients with interstitial cystitis in the city of Helsinki". This superb, brief report from Finland surveyed all diagnosed cases in a population approaching 1 million. The prevalence of the disease in women was 18.1 per 100,000. The joint prevalence in both sexes was 10.6 cases per 100,000. The annual incidence of new female cases was 1.2 per 100,000. Severe cases accounted for about 10% of the total. Ten percent of cases were in men. The disease onset was generally subacute rather than insidious, and full development of the classic symptom complex occurred over a relatively short time. IC does not progress continuously but usually reaches its final stage rapidly (within 5 years in the study by Koziol and colleagues) and then continues without significant change in symptomatology. Subsequent major deterioration was found by Oravisto to be unusual. The duration of symptoms before diagnosis was 3 to 5 years in the Finnish study. Analogous figures in a classic American paper a quarter of a century earlier were 7 to 12 years.
Another early population study, this in the United States, first demonstrated the potential extent of what had been considered a very rare disease. The following population groups were surveyed: (1) random survey of 127 board-certified urologists, (2) 64 IC patients selected by the surveyed urologists and divided between the last patient with IC seen and the last patient with IC diagnosed, (3) 904 female patients belonging to the Interstitial Cystitis Association, and (4) a random phone survey of 119 persons from the U.S. population. This 1987 study reached the following conclusions:
-
There were 43,500 to 90,000 diagnosed cases of IC in the United States (twice the Finnish prevalence).
-
Up to a fivefold increase in IC prevalence occurred if all patients with painful bladder and sterile urine had been given the diagnosis, yielding up to a half million possible cases in the United States.
- Median age at onset is 40 years.
- Late deterioration in symptoms is unusual.
- There is a 50% temporary spontaneous remission rate, with a mean duration of 8 months.
- The incidence of childhood bladder problems is 10 times higher in IC patients versus controls.
- The incidence of a history of urinary tract infection is twice that of controls.
-
Fourteen percent of IC patients were Jewish (15% in the sample of Koziol [1994 ]) versus 3% who were Jewish in a general population sample.
- IC patients have a lower quality of life than dialysis patients.
- Costs including lost economic production, in 1987, were $427 million.
Other population studies followed. Jones and Nyberg obtained their data from a self-report of a previous diagnosis of IC in the 1989 National Household Interview Survey. The survey estimated that 0.5% of the population, or more than 1 million people in the United States, reported having a diagnosis of IC. There was no verification of this self-report by medical records. Bade and associates did a physician questionnaire–based survey in the Netherlands yielding an overall prevalence of 8 to 16 per 100,000 females, with the diagnosis heavily dependent on pathology and presence of mast cells. This prevalence in females compares to 4.5 per 100,00 in Japan. The Nurses Health Study I and II showed a prevalence of IC between 52 and 67 per 100,000 in the United States, twice the prevalence in the study of Held and associates and threefold greater than the study of Bade and coworkers. It improved on previous studies by using a large sample derived from a general population and careful ascertainment of the diagnosis. If the 6.4% confirmation rate of their study were applied to the Jones and associates' National Health Interview Survey data, the prevalence estimates of the two studies would be nearly identical.
Leppilahti and colleagues and Miller and associates used the O'Leary-Sant IC Symptom and Problem Index (never validated for making a diagnosis per se) to select persons with IC symptoms from the Finnish population register. They calculated an incidence, based on an index score of 12 or greater, of 450/100,000. Roberts , using a physician diagnosis as the arbiter of IC, found annual incidence in Olmsted County, Minnesota, of 1.6 per 100,000 in women and 0.6 per 100,000 in men, a figure remarkably similar to the findings of Oravisto in Helsinki. The cumulative prevalence by age older than 80 years in the Minnesota study was 114 per 100,000, a figure comparable to that in the Nurses Health Study if one takes into account the younger age group in the Curhan data. Clemens and associates calculated a prevalence of diagnosed disease in a managed care population of 197 per 100,000 women and 41 per 100,000 men, but when the diagnosis was tested by eliminating those who had not been evaluated with endoscopy or in whom exclusionary conditions existed, the numbers dropped considerably.
Whether the considerable variability in prevalence in studies within the United States and around the world is related to methodology or true differences in incidence is an important question yet to be answered. Familial occurrence of PBS/IC has been reported. A hereditary aspect to incidence has been suggested by Warren and associate in a pioneering study. They found that adult female first-degree relatives of patients with IC may have a prevalence of IC 17 times that found in the general population. This, together with previously reported evidence showing a greater concordance of IC among monozygotic than dizygotic twins suggests but does not prove a genetic susceptibility to IC that could partially explain the discord in prevalence rates in different populations.
The ICDB cohort of patients has been carefully studied, and the findings seem to bear out those of other epidemiologic surveys. Patterns of change in symptoms with time suggest regression to the mean and an intervention effect associated with the increased follow-up and care of cohort participants. Although all symptoms fluctuated, there was no evidence of significant long-term change in overall disease severity. The data suggest that PBS/IC is a chronic disease and no current treatments have a significant impact on symptoms over time in the majority of patients. Quality of life studies suggest that PBS/IC patients are six times more likely than individuals in the general population to cut down on work time owing to health problems but only half as likely to do so as patients with arthritis. There is an associated high incidence of comorbidity, including depression, chronic pain, and anxiety and overall mental health. There seems to be no effect on pregnancy outcomes.
Most studies show a female-to-male preponderance of 5:1 or greater. In the absence of a validated marker, it is often difficult to distinguish PBS/IC from the chronic pelvic pain syndrome (nonbacterial prostatitis, prostatodynia) that affects males, and the percentage of men with PBS/IC may actually be higher. Men tend to be diagnosed at an older age and have a higher percentage of Hunner's ulcer in the case series reported.
Associated Diseases
Knowledge of associated diseases is relevant for the clues it engenders with regard to etiology and possible treatment of this enigmatic pain syndrome. In a case-control study, Erickson and associates found that patients with IC had higher scores than control subjects for pelvic discomfort, backache, dizziness, chest pain, aches in joints, abdominal cramps, nausea, palpitations, and headache. Buffington theorizes that a common stress response pattern of increased sympathetic nervous system function in the absence of comparable activation of the hypothalamic-pituitary-adrenal axis may account for some of these related symptoms. It has recently been hypothesized that panic disorder may sometimes be a part of a familial syndrome that includes IC, thyroid disorders, and other disorders of possible autonomic or neuromuscular control.
KEY POINTS: EPIDEMIOLOGY |
Prevalence estimates per 100,000 persons (see text for details):
An accurate country-by-country determination of PBS/IC prevalence and incidence is difficult to perform at the present time. Until specific diagnostic markers are verified and/or a set of agreed-upon diagnostic criteria based on well-designed published data are established, it seems most appropriate to use a more inclusive symptom specific definition of PBS/IC to permit an assessment of the population burden. Specifically, a validated questionnaire that can be administered in person or by telephone and that has been compared with a gold standard (disease marker in the future; expert opinion and diagnosis at present) is required to estimate within a given range the true extent of disease in populations around the world. |
Newly diagnosed patients are most concerned with the possibility that PBS/IC could be a forerunner of bladder carcinoma. No reports have ever documented a relationship to suggest that IC is a premalignant lesion. Utz and Zincke discovered bladder cancer in 12 of 53 men treated for IC at the Mayo Clinic. Three of 224 women were eventually diagnosed with bladder cancer. Four years later additional cases were reported. Tissot and associates reported 1% of 600 patients previously diagnosed as having IC were found to have transitional cell carcinoma as the cause of symptoms. Somewhat ominously, 2 of these patients had no hematuria. In all patients, irritative symptoms resolved after treatment of the malignancy. From these experiences has come the dictum that all patients with presumed IC should undergo cystoscopy, urine cytology, and bladder biopsy of any suspicious lesion to be sure that a bladder carcinoma is not masquerading as PBS/IC. It would seem that in the absence of microhematuria, and with a negative cytology, the risk of missing a cancer is negligible, but not zero. There is no evidence that PBS/IC itself is associated with a higher risk of bladder cancer or of transitions to cancer over time.
A large-scale survey of 6783 individuals diagnosed by their physicians as having IC studied the incidence of associated disease in this population (Fig. 2). Data from the 2405 responders were validated by comparison with 277 nonresponders. Allergies were the most common association, with over 40% affected. Allergy was also the primary association in Hand's study. Thirty percent of patients had a diagnosis of irritable bowel syndrome, a finding confirming that of Koziol. Altered visceral sensation has been implicated in irritable bowel syndrome in that these patients experience intestinal pain at intestinal-gas volumes that are lower than those that cause pain in healthy persons, strikingly similar to the pain on bladder distention in IC.
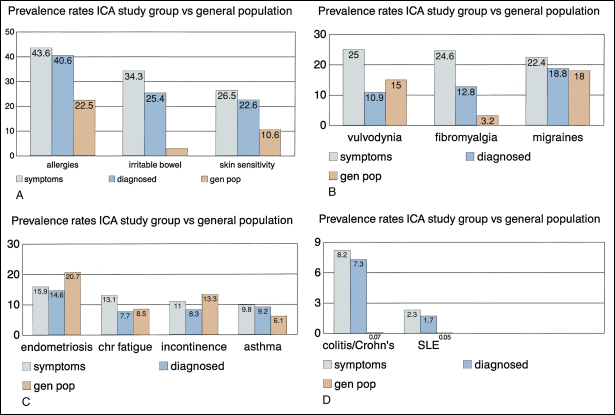 |
| Figure 2. A to D, Comparison of disease prevalence rates between the Interstitial Cystitis Association (ICA) study group patients who report symptoms of a disorder, who have been diagnosed with a disorder, and the general population. SLE, systemic lupus erythematosus. |
Fibromyalgia, another disorder frequently considered functional because no specific structural or biochemical cause has been found, is also overrepresented in the IC population. This is a painful, nonarticular condition predominantly involving muscles; it is the most common cause of chronic, widespread musculoskeletal pain. It is typically associated with persistent fatigue, nonrefreshing sleep, and generalized stiffness. As in IC, women are affected at least 10 times more often than men. The association is intriguing because both conditions have nearly identical demographic features, modulating factors, associated symptoms, and response to tricyclic compounds.
Vulvodynia, migraine headaches, endometriosis, chronic fatigue syndrome, incontinence, and asthma had similar prevalence as in the general population. Several publications have noted an association between IC and systemic lupus erythematosus (SLE). The question has always been as to whether the bladder symptoms represent an association of these two disease processes or rather are a manifestation of lupus involvement of the bladder or even a myelopathy with involvement of the sacral cord in a small group of these patients. The beneficial response of the cystitis of SLE to corticosteroids tends to support the latter view. No association with discoid lupus has been demonstrated. Although the actual numbers are small, the Alagiri study demonstrated a 30 to 50 times greater incidence of SLE in the IC group compared with the general population. Overall, the incidence of collagen vascular disease in the IC population is low. Parsons found only 2 of 225 consecutive IC patients to have a history of autoimmune disorder.
Inflammatory bowel disease was found in over 7% of the IC population Alagiri studied, a figure 100 times higher than in the general population. While unexplained at this time, abnormal leukocyte activity has been implicated in both conditions.
Another mysterious disorder that has been associated with IC is focal vulvitis. Vulvar vestibulitis syndrome is a constellation of symptoms and findings involving and limited to the vulvar vestibule consisting of (1) severe pain on vestibular touch to attempted vaginal entry, (2) tenderness to pressure localized within the vulvar vestibule, and (3) physical findings confined to vulvar erythema of various degrees. McCormack reported on 36 patients with focal vulvitis, 11 of whom also had IC. Fitzpatrick and associates have added three more cases. The concordance of these noninfectious inflammatory syndromes involving the tissues derived from the embryonic urogenital sinus and the similarity of the demographics argue for a common etiology.
An association has been reported between IC and Sjögren's syndrome, an autoimmune exocrinopathy with a female preponderance manifested by dry eyes, dry mouth, and arthritis but that can also include fever, dryness, and gastrointestinal and lung problems. Van De Merwe and coworkers investigated 10 IC patients for the presence of Sjögren's syndrome. Two patients had both the keratoconjunctivitis sicca and focal lymphocytic sialoadenitis, allowing a primary diagnosis of Sjogren's syndrome. Only 2 patients had neither finding. They later reported an incidence of 28% of Sjögren's syndrome in patients with IC. The incidence of symptoms of PBS/IC in patients with Sjögren's syndrome has been estimated to be up to 5%.
A negative correlation with diabetes has been noted.
Further epidemiologic studies are warranted, because the epidemiology of this disorder may ultimately yield as many clues into etiology and treatment as other avenues of research.
KEY POINTS: ASSOCIATED DISORDERS |
|
ETIOLOGY
It is likely that PBS/IC has a multifactorial etiology that may act predominantly through one or more pathways resulting in the typical symptom-complex (Fig. 3). There are an abundance of theories regarding its pathogenesis, but confirmatory evidence gleaned from clinical practice has proven sparse. Among numerous proposals that shall be further explored in this section are "leaky epithelium", mast cell activation, and neurogenic inflammation, or some combination of these and other factors leading to a self-perpetuating process resulting in chronic bladder pain and voiding dysfunction.
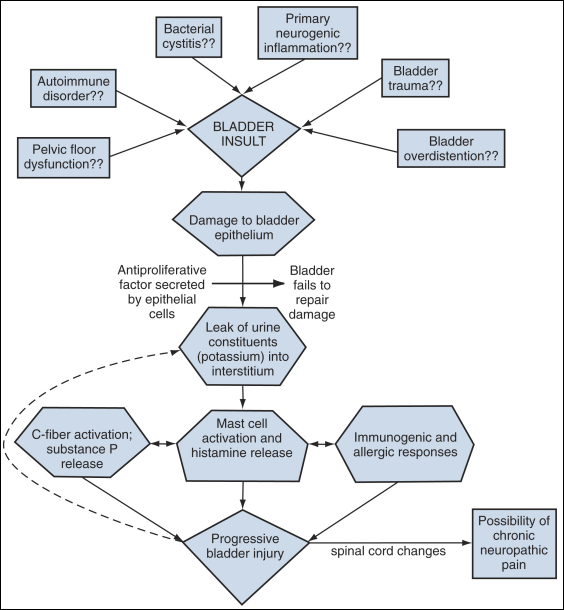 |
| Figure 3. Hypothesis for etiologic cascade of painful bladder syndrome/interstitial cystitis. |
Animal Models
Until recently, lacking an easily available animal model of the naturally occurring disease, researchers have had to devise animal models to study isolated symptoms of PBS/IC, hoping to uncover the root causes of the symptomatology. Bullock and associates reported a mouse model in which bladder inflammation could be induced by the injection of syngeneic bladder antigen. While demonstrating that a component in the Balb/cAN mouse is capable of inducing a bladder-specific, adoptively transferable, cell-mediated autoimmune response that exhibits many characteristics of clinical IC, the model became difficult to reproduce.
A guinea pig model was used in which a solution was instilled containing a protein to which the animal had been previously immunized and which resulted in bladder inflammation, and a rat model was used of allergic cystitis using a local challenge of ovalbumin in previously sensitized rats. Changes in the rat model were dependent on mast cell degranulation and activation of sensory C fibers.
Ghoniem and coworkers studied four female African green monkeys challenged with intravesical acetone. Not surprisingly, they exhibited symptoms of painful bladder syndrome. Rivas and associates performed similar experiments using dilute hydrochloric acid in a rat model. A rat model for neurogenic cystitis using pseudorabies virus demonstrated that inflammatory changes in the spinal cord can result in dramatic, neurogenically mediated changes in the bladder.
The problem with all of these animal models relates to whether they mirror the human disease to any great extent. Buffington has described what appears to be a naturally occurring animal model of PBS/IC. Two thirds of cats with lower urinary tract disease have sterile urine and no evidence of other urinary tract disorders. A portion of these cats experience frequency and urgency of urination, pain, and bladder inflammation (Fig. 4). Glomerulations have been observed in the bladders of these animals. GP-51, a glycosaminoglycan (GAG) commonly found in the surface mucin covering the mucosa of the normal human bladder and decreased in IC, shows a decreased expression in cats with this symptom-complex, originally termed feline urologic syndrome. Bladder Aδ afferents in these cats are more sensitive to pressure changes than are afferents in normal cats. These cats also demonstrate an increase in baseline nitric oxide production in smooth muscle and mucosal strips when compared with healthy cats with evidence of altered mucosal barrier function.
 |
| Figure 4. Photograph of cat suffering from feline interstitial cystitis. |
Buffington and associates now refer to this disorder as feline interstitial cystitis. It is associated with urinary urgency, frequency, and pain with sterile urine, bladder mastocytosis, increased histamine excretion, increased bladder permeability, decreased urinary GAG excretion, and increased plasma norepinephrine concentrations.
Although animal models can yield clues to etiology, all theories must ultimately be tested in humans with the disease.
Infection
Often, a diagnosis of PBS/IC is made only after a patient has been seen by a number of physicians and treated with antibiotics for presumed urinary tract infection without resolution of symptoms. The symptom-complex looks to the patient and physician like an infectious process. The epidemiology of urinary tract infection and its predominance in women mirror the IC data. The acute to subacute onset in many patients has fascinated clinicians, who often associate an insidious onset with a chronic condition such as PBS/IC.
Reverse logic led some to suspect that antibiotics may be instrumental in causing IC. Most patients have been treated with antibiotics once or several times before the diagnosis is made. Numerous antibiotics, primarily in the penicillin family, can induce a cystitis, but no evidence has ever been documented that these antibiotics or the supposedly "surface active" nitrofurantoins or tetracyclines have any involvement in pathogenesis.
To determine whether there is an infectious cause of IC certain procedures are necessary. Not just urine but bladder epithelium as well must be cultured for appropriate microorganisms, including bacteria, viruses, and fungi. Because some organisms might be culturable yet fastidious, special culture techniques should be used. Because some organisms in urine or tissue might be viable but nonculturable, specific nonculture techniques for discovery and identification should be employed. Most important, the same procedures must be carried out in a control population.
Attempts to show an infectious etiology go back to the dawn of the disease, but the case has never been a strong one. Hunner originally proposed that IC resulted from chronic bacterial infection of the bladder wall secondary to hematogenous dissemination. Harn and associates proposed a relationship between IC and streptococcal and post-streptococcal inflammation. They produced a progressive chronic inflammation in rabbit bladders by injecting small numbers of Streptococcus pyogenes in the bladder wall. The discovery that Helicobacter pylori is related to the pathogenesis of chronic atrophic gastritis and peptic ulcer disease and that antibiotic treatment can heal ulceration has continued to focus attention by researchers in IC on the possibility that an infectious etiology is not only reasonable but will ultimately be found. Studies of H. pylori itself have failed to demonstrate an association with IC. Wilkins and coworkers found bacteria in catheterized urine specimens and/or bladder biopsies in 12 of 20 patients with IC. However, eight of the isolates were fastidious bacteria, Gardnerella vaginalis, and Lactobacillus species and no control subjects were included in the study.
Negative studies far outnumber the positive ones. Hanash and Pool performed viral, bacterial, and fungal studies on 30 IC patients and failed to substantiate an infectious etiology. Hedelin and colleagues found only 3 of 19 IC patients to have urine cultures positive for Ureaplasma urealyticum and indirect hemagglutination antibodies to Mycoplasma hominis to be no greater than in controls. Potts and associates cultured U. urealyticum in 22 of 48 patients with "chronic urinary symptoms" and had great success in these patients (none of whom had established IC) with short courses of commonly prescribed antibiotics. Given the history of empirical use of antibiotics in the vast majority of IC patients, it is doubtful if this group represents even a small percentage of the IC-diagnosed population. Nevertheless, it illustrates that IC is a diagnosis of exclusion, and urine culture is critical while an empirical short course of antibiotics is certainly reasonable if the patient has not already been treated for presumed infection. Empirical use of doxycycline at a dose of 100 mg twice daily for 2 weeks followed by 100 mg daily for 2 weeks has been successfully used in this manner.
The development of highly sensitive, rapid, and specific molecular methods of identifying infectious agents by the direct detection of DNA or RNA sequences unique to a particular organism resulted in a flurry of activity into the search for a responsible virus or microorganism. Hampson and coworkers could find no evidence of mycobacterial involvement in eight cases of IC using DNA probes. Haarala and coworkers confirmed an absence of bacterial DNA in the bladder of 11 IC patients with no documented history of urinary tract infection. Hukkanen and colleagues reported an absence of adenovirus and BK virus genomes in urinary bladder biopsies of IC patients. Domingue and associates provocative finding of the presence of bacterial 16S rRNA genes in bladder biopsies from 29% of IC patients but not from control bladders, and their discovery of 0.22-mm filterable forms in culture of biopsy tissue from 14 of 14 IC patients and none of 15 control subjects has never been confirmed or repeated.
A preliminary study found a statistically significant increase in urine PCR to Chlamydia pneumoniae major outer membrane protein gene in patients with IC as compared with controls. Other studies have shown that similar percentages of both IC and control patient populations have nonculturable bacteria in their bladder on the basis of polymerase chain reaction studies of bladder biopsy specimens. The spirochete Borrelia burgdorferi has been found in the bladder biopsies and urine of patients with Lyme disease and can cause frequency, urgency, and nocturia. DNA studies have failed to show a role for Borrelia in IC.
The role of infection in the pathogenesis of IC remains a mystery. At this time there are little data to support the role of an infectious etiology but investigators keep returning to an infectious theory. New insights into the mechanisms by which bacteria adhere, grow, and persist in association with host tissue and form intracellular pods capable of subverting host defense mechanisms and allowing replication within epithelial cells lay the foundation for a possible role of infection in initiating the PBS/IC pathologic cascade. The University of Maryland group proposed a model of IC in which bladder epithelial damage such as that caused by bacterial cystitis may be the first step leading to a low-level inflammatory response we call IC. Domingue and Ghoniem write that "It is logical to suggest that even if organisms are not causative agents, their presence may lead to immune and host-cell responses that could initiate or exacerbate an inflammatory state".
If infection does play a role, it would be predicted that appropriate treatments to minimize microbial presence in the tissue would significantly improve the morbidity associated with IC. Durier's incredible series purporting to cure 27 of 27 IC patients with the use of up to five sequential antibiotics covering the anaerobic spectrum has never been duplicated. Warren and colleagues' prospective, double-blind, placebo-controlled, randomized trial of 50 patients may well prove the end of long-term empirical antibiotic treatment in established IC. Eighteen weeks of placebo or antibiotics (sequential doxycycline, erythromycin, metronidazole, clindamycin, amoxicillin, and ciprofloxacin for 3 weeks each) were administered. Most patients guessed the arm to which they were assigned. Of the 25 patients in the active arm, 80% had new non-urinary symptoms perceived as side effects. There was minimal improvement in some patients associated with the active arm of the study, but the conclusion that intensive antibiotics do not represent a major advance in therapy for IC seems well justified.
KEY POINT: ETIOLOGY (INFECTION) |
| Although the concept that a urinary tract infection may trigger IC in some patients is appealing, it is unlikely that active infection is involved in the ongoing pathologic process or that antibiotics have a role to play in treatment. |
Autoimmunity / Inflammation
Immune/neuroimmune mechanisms may have an important role in the pathogenesis of PBS/IC. Excessive release of sensory nerve neurotransmitters and mast cell inflammatory mediators is thought to be responsible for the development and propagation of symptoms. Inflammation results in altered nerve growth factor content of the bladder and in morphologic changes in sensory and motor neurons innervating the bladder. Such neuroplasticity may be a possible explanation for the association of bladder inflammation with long-term symptoms and pain after inflammation subsides.
For many years the possibility that IC may represent some type of autoimmune disorder has been considered. Narrowly defined, autoimmune diseases are clinical syndromes caused by the activation of T cells or B cells, or both, in the absence of an ongoing infection or other discernible cause. To establish a disease as autoimmune, three types of evidence can be marshaled: (1) direct evidence from transfer of pathogenic antibody or pathogenic T cells, (2) indirect evidence based on reproduction of the autoimmune disease in experimental animals, and (3) circumstantial evidence from clinical clues. Circumstantial evidence would include (1) association with other autoimmune diseases in the same individual or same family; (2) lymphocytic infiltration of target organ; (3) statistical association with a particular major histocompatibility complex haplotype; and (4) favorable response to immunosuppression.
Circumstantial evidence by itself cannot define an autoimmune disease, and at this point the case for autoimmunity in IC is far from clear. Three different possibilities exist: (1) IC is caused by a direct autoimmune attack on the bladder, (2) some of the autoimmune symptoms and pathology of IC arise indirectly as a result of tissue destruction and inflammation from other causes, and (3) autoimmune phenomena in IC patients are coincident and unrelated to the disease.
Silk found bladder antibodies in 9 of 20 IC patients and none in 35 pathologic or normal control patients. Gordon and associates found anti-bladder antibodies present in biopsy specimens from 6 of 8 IC patients and in 3 of 5 control patients. No control patient demonstrated antibodies in the muscle, whereas 3 of 5 IC patients with muscle in the biopsy did so. Jokinen and associates looked at sera from 33 IC patients and found 28 with an antinuclear antibody (ANA) titer of 1:10 or greater, but no bladder-specific antibodies were detected with immunofluorescence. There was poor correlation between ANA titers and symptom severity. Jokinen and associates noted that elevated antibody titers against cell nuclei and crude kidney homogenate decreased within 12 months after cystectomy in 3 IC patients. All of this provided hints that IC could fall into the autoimmune group of diseases.
Oravisto summarized the world literature on this idea in 1980, concluding that the chronic course of disease, the absence of infection, the pathologic findings, the occurrence of antinuclear antibodies, and the reported responses to corticosteroids at that time provided strong circumstantial evidence of autoimmunity. He discounted the paucity of activated lymphocytes which speak against an autoimmune process. Studies on autoantibodies in PBS/IC have shown that these mainly consist of antinuclear antibodies similar to the autoantibody profiles in some systemic diseases such as Sjögren's syndrome, well known to be of autoimmune origin. Mattila presented evidence of immune deposits in the bladder vascular walls in 33 of 47 IC patients. Studying sera from 41 patients with IC, he concluded that the classical pathway activation of the complement system was involved, supporting the possibility that a chronic local immunologic process was indeed occurring. The autoantibodies tested were found to be directed against cytoskeletal intermediate filaments. Because the autoantibodies have to gain access to intracellular structures to cause in vivo deposits, primary tissue injury of unknown etiology has to be postulated.
Anderson and colleagues studied 26 patients with IC and compared them with a control group of similar age and sex with other urologic complaints. They performed a standard autoimmune profile and looked for specific antibodies to normal human bladder in the serum. Sixty-five percent of IC patients and 36% of controls demonstrated non–organ-specific antibodies; 40% of IC patients had ANAs; 75% of IC patients and 40% of controls had anti-bladder antibodies present in the serum. There was no increase in immunoglobulin deposition in the bladder epithelium in IC patients versus controls. Although IC patients demonstrated a nonspecific increase in antibody formation, this was not significantly different from a similar group of other urologic patients. The lack of specificity indicates the immunologic findings are likely secondary to inflammation rather than a primary etiology.
In a study looking for active immune cellular deposition in IC patients, no statistically significant difference between controls and nonulcerative IC patients was identified. In contrast, the ulcerative IC group had focal sheets of plasma cells, aggregates of T cells, B cell nodules, a decreased or normal helper-to-suppressor cell ratio, and suppressor cytotoxic cells in germinal centers. Flow cytometry analysis of peripheral blood lymphocyte subsets showed increased numbers of secretory Ig–positive B cells and activated lymphocytes in the nonulcerative group and increased numbers of secretory Ig–positive cells and activated lymphocytes in the ulcerative group. These results may suggest a partial role for an immune mechanism in IC. Erickson and coworkers have also noted a major difference in inflammatory cell types as well as clinical features in IC patients with severe inflammation, suggesting two different patient groups with two different underlying pathophysiologies.
Hanno and colleagues found CD4 cell predominance in all layers of the bladder in IC patients. Christmas reported increased numbers of CD4+ and CD8+ T cells in bladder biopsies from patients with IC and bacterial cystitis as compared with controls. These T cells were present in the urothelium and submucosa but not in the detrusor. Control bladder tissue demonstrated only CD8 cells in the urothelium and both CD4+ and CD8+ cells in the submucosa. The number of plasma cells was significantly greater in IC patients than in normal controls and controls with bacterial cystitis.
MacDermott and colleagues found a normal distribution of peripheral blood lymphocytes in IC patients, a finding not supportive of an autoimmune mechanism in the disease. The lamina propria showed a predominance of CD4 (helper T cells) lymphocytes over CD8 cells in both IC and other cystitis patients. The same pattern was seen in the epithelium of patients with bacterial or mechanical cystitis, but patients with IC had a predominance of CD8 lymphocytes in the urothelium — identical to control subjects. The findings suggest that the urothelium is not involved in the inflammatory reaction, as is the lamina propria, making the urothelium an unlikely source for the initiating factor.
Miller and coworkers investigated the function of peripheral blood lymphocytes from nonulcerative IC patients, testing the proliferative response and cytokine production of T cells to nonspecific mitogenic stimulation and the proliferative response of T cells to urine components. Proliferation and cytokine production after mitogen stimulation were the same for control subjects and IC patients. Moreover, no immunologic response to IC urine by autologous peripheral blood lymphocytes in in-vitro assays was observed. Their findings cast doubt on theories suggesting that IC is an autoimmune disease.
Numerous inflammatory mediators have been studied with regard to their relation to IC. Patients with PBS/IC exhibit varying degrees of inflammation that can separate them into clusters. Bladder inflammation in IC is categorized by elevated urinary interleukin-6 and activation of the kallikrein-kinin system. The absence of urinary interleukin-1β in IC argues against an immunologic or autoimmune etiology of the disorder. Neurogenic inflammation may play a role in the etiology, because long-term exposure of afferent nerve terminals to inflammatory mediators can alter ion channels and result in bladder hyperalgesia. Substance P itself does not seem to be the single initiator of inflammation in the bladder, and its blockade does not protect the bladder in animal models from inflammatory responses. Urinary nitric oxide synthase (NOS) activity is known to be elevated in patients with urinary infection and thought to play a role in the bladder's response to infection and in the inflammatory process that follows infection. The finding that urinary NOS activity is decreased in IC patients has puzzled researchers but could explain the reduction in functional bladder capacity associated with the disorder.
Urothelial cell activation in IC may result in aberrant immune responses and immune activation within the bladder wall that could relate to pathogenesis of the disease but might not reflect initiating etiology. It has been proposed that inflammatory and/or immune responses in IC could be exacerbated by persistent activation of the nuclear factor (NF)-kB. Angiogenic factors such as platelet-derived endothelial cell growth factor/thymidine phosphorylase and transforming growth factor-β may be involved in the inflammatory process to induce painful symptoms in patients with IC or bladder carcinoma.
The exact role of autoimmunity in IC remains controversial. Suplatast tosilate, a new immunoregulator, has shown efficacy in a small, uncontrolled IC study in which improvements in symptoms and bladder capacity were correlated with changes in autoimmune parameters. Although the immune system remains a target for therapy, no clear indication of a primary role for autoimmunity as the cause of IC has been observed.
Mast Cell Involvement
Although mast cells are thought of primarily in the context of allergic disorders, and certain acute inflammatory responses, these cells have also been implicated in biologic responses as diverse as angiogenesis and wound healing, bone remodeling, peptic ulcer disease, atherosclerosis, and reactions to neoplasms. Mast cells remain one of the most enigmatic cells in the body. They secrete significant amounts of numerous proinflammatory mediators that contribute to a number of chronic inflammatory conditions, including stress-induced intestinal ulceration, rheumatoid arthritis, scleroderma, and Crohn's disease. They have been described even among the lowest order of animals, having been discovered in the frog mesentery over 100 years ago. Their raison d'etre may be for initiating and coordinating the host's inflammatory and immune responses against microbial pathogens. Recently they have been implicated in a range of neuroinflammatory diseases, especially those worsened by stress. These include multiple sclerosis, migraines, inflammatory arthritis, atopic dermatitis, coronary inflammation, irritable bowel syndrome, and PBS/IC. They may be activated through their Fc receptors by immunoglobulins other than IgE, as well as by anaphylatoxins, neuropeptides, and cytokines to secrete mediators selectively without overt degranulation.
Mast cells have frequently been reported to be associated with IC, both as a pathogenetic mechanism and as a pathognomonic marker. The association of bladder mastocytosis, IC, and irritable bowel syndrome and chronic urticaria is intriguing. Evidence of their importance is mounting, suggesting that they may serve as the final common pathway through which the symptomatic condition is expressed. Mast cells produce, among other compounds, histamine. Histamine release in tissue causes pain, hyperemia, and fibrosis, all notable features of IC.
Simmons was the first to suggest mast cells as a cause of IC. Contribution of mast cells to the cellular infiltrate in IC (Fig. 5) has been shown to vary from about 20% in nonulcer IC patients to 65% in patients with ulceration. Mast cells participate in allergic reactions (hypersensitivity type I) during which IgE antibody is synthesized in response to specific antigens. IgE binds to mast cell receptors, and antigen binds to the IgE, leading to degranulation. Other triggers of mast cell secretion include acetylcholine, anaphylatoxins, cationic peptides such as substance P, chemicals, contrast agents, cytokines, opioids, antihistamines, exercise, hormones, viruses, and bacterial toxins. Mast cells promote infiltration of neutrophils, T and B lymphocytes, monocytes, and eosinophils. T lymphocytes secrete substances capable of activating mast cells, thus perpetuating the cycle of inflammation.
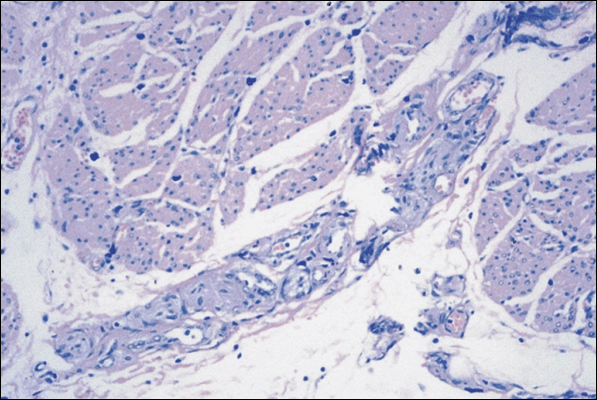
|
| Figure 5. Giemsa stain shows detrusor mastocytosis and nerve hypertrophy in interstitial cystitis. (Original magnification, x400.) |
Since the presence of mast cells within the bladder wall was first recognized, numerous investigators have tried to determine whether there is an increase in the number of mast cells in the bladder of patients with IC, or differences in their location or functional state. An increase in urothelial mast cells appears to be part of the generalized inflammatory cell reaction regardless of etiology and not a specific feature of IC, whereas the presence of increased numbers of mast cells in the detrusor is more specific for IC. However, one study did report detrusor mastocytosis in 64% of IC patients and 80% of a control group with other urologic disease, with no statistically significant difference between the mean number of detrusor mast cells in the two groups.
Aldenborg and associates reported that mast cells are found predominantly in the detrusor muscle in patients with classic IC, but there is also a secondary population of mast cells in the lamina propria and the bladder epithelium, with staining characteristics distinct from those in the detrusor. None of these epithelial mast cells were found in controls. These findings were interpreted to indicate a transepithelial migration of mast cells in patients with IC. This second population of mast cells does not appear to be involved in the nonulcer type of IC. This mucosal population of mast cells can also differ from the mast cells found in deeper tissues in physiologic responses and release of secretory products. The "mucosal mast cells" are susceptible to aldehyde fixation and require special fixation and staining techniques for proper demonstration. Detrusor mast cells are not susceptible to fixation techniques. Recent studies have shown that although all human mast cells contain the proteinase tryptase, there is a population of mast cells that also contain the proteinase chymase. The mast cell expansion in IC involves both types. Mast cell activation is far more pronounced in the ulcerative form, which in addition displays prominent inflammation, in contrast to nonulcer IC, where it is sparse. Thus, the basic pathologic processes may differ. Because activated mast cells lose their histologically identifiable granules once degranulation occurs, estimates of mast cell density using standard histologic techniques may underestimate mast cell numbers.
Electron microscopy has confirmed that mast cells in IC are more likely to be degranulated or activated than those found in other conditions. In at least a subpopulation of IC patients, this may be explained by increased stimulation of mast cells by stem cell factor. A chronic exposure of detrusor muscle to histamine in IC patients is suggested by the finding that there is an impairment of the direct contractile response to histamine in detrusor muscle affected by IC in comparison to control detrusor, suggesting a receptor desensitization. The clinical relationship between an increased number of mast cells and symptoms of IC has not been definitively established. Some studies have found no correlation. Although mast cell infiltration in intestinal segments used for augmentation has been associated with pain and failure of the procedure, others have shown that mast cell infiltration in intestine used in the urinary tract is the norm and not pathologic.
Many of the substances that have been shown to induce mast cell secretion are released from neurons that innervate the organ containing the mast cells. The capsaicin-sensitive sensory neurons that innervate the bladder are thought to have a dual "sensory-efferent" function, in which an axon reflex–induced release of neuropeptides results in local inflammation. Hand reported an increase in the submucosal nerve density in IC, a phenomenon confirmed by Christmas and colleagues , who showed an increase in nerve fiber proliferation in IC but not in patients with bacterial or lupus cystitis. Increased innervation by nerves releasing substances affecting mast cells could lead to increased mast cell secretion. Among these substances is acetylcholine. Mast cells can be stimulated by cholinergic agonists to secrete serotonin. Substance P–containing fibers have been found to be increased in bladders from IC patients and are found adjacent to mast cells. In mice, mast cells modulate the inflammatory response of the bladder to substance P and to E. coli lipopolysaccharide.
An increase in adrenergic but not cholinergic nerves in IC patients as compared with controls has been reported. These researchers also found increased numbers of neurons staining for vasoactive intestinal polypeptide and neuropeptide Y (NPY), both of which are associated with sympathetic nerves. Studies in rats have revealed that psychological stress can activate bladder mast cells via the action of sensory neuropeptides. Diurnal cortical variations have been associated with symptom levels in PBS/IC, and the mast cell may represent a pathway for stress to be reflected in bladder symptomatology.
Mast cells can alter their environment by regulating tissue gene expression. The finding of increased synthesis of urinary leukotriene E4 in patients with IC and detrusor mastocytosis when compared with healthy control subjects suggests that cysteinyl-containing leukotrienes are involved in the inflammatory reaction observed in the urinary bladder of patients with IC and may be produced from tissue mast cells in the bladder wall or from macrophages .
Could mast cell products be useful in diagnosing IC? They are not specific for PBS/IC and are increased in bladder carcinoma. Elevated histamine levels have been found in bladder biopsies of IC patients as well as from bladder washings. Holm-Bentzen and coworkers reported a significantly elevated urinary excretion of 1,4-methylimidazole acetic acid, the major metabolite of histamine. Others have found no differences between IC and controls in random spot tests of urinary histamine. Levels were elevated after hydrodistention in IC patients but not in control subjects, a possible consequence of hydrodistention and resultant mast cell degranulation. El Mansoury and associates found increased methylhistamine, a histamine metabolite, in spot and 24-hour urine samples from IC patients as compared with control subjects. While such an increase could still be interpreted as indicating a systemic rather than a bladder syndrome, subsequent findings of elevated mast cell tryptase in the urine of IC patients could only come from the bladder. Erickson and colleagues reported that urine methylhistamine is not useful as an objective marker of response to bladder distention or as a predictor of response to distention or as a substitute for bladder biopsy to determine mast cell counts.
The realization that mast cells are associated with the syndrome of IC by no means diminishes the other multiple theories of causation. Their very presence could be related to injury from any of the proposed theories of etiology, and degranulation could likewise reflect a final common pathway resulting in pain and frequency from multiple causes. Rickard and Lagunoff proposed, based on results with mast cell granules and epithelial cells in tissue culture, that mast cells could contribute to failure of epithelialization of the bladder surface following injury by two potential mechanisms: (1) inhibition of epithelial cell replication and (2) interference with epithelial cell spreading, thus resulting in the "leaky epithelium" found in some patients. Mast cells may actually be the mediator through which female hormones play a role, accounting for the 10:1 female to male preponderance of the disease. Estradiol augments the secretion of mast cell histamine in response to substance P. It has been proposed that the symptoms of IC may depend on an imbalance of the relative number of estrogen receptors to progesterone receptors on bladder mast cells.
KEY POINT: ETIOLOGY (MAST CELL INVOLVEMENT) |
| Mast cells are strategically localized in the urinary bladder close to blood vessels, lymphatics, nerves and detrusor smooth muscle ( Saban et al, 1997 ). Studies strongly suggest that IC is a syndrome with neural, immune, and endocrine components in which activated mast cells play a central, although not primary, pathogenetic role in many patients ( Elbadawi and Light 1996 ; Filippou et al, 1999 ). |
Bladder Glycosaminoglycan Layer and Epithelial Permeability
Until the early 1970s, most investigators thought that the major barrier to free flow of urinary constituents was at the level of the epithelial cells. Tight junctions between urothelial cells, specialized "umbrella cells" lining the surface, and direct bactericidal activity of the vesical mucosa were thought capable of defense of the internal milieu from bacteria, molecules, and ions in the urine". Staehelin and colleagues proposed that lipid and other hydrophobically bonded materials were important in any barrier to permeability in the luminal membrane because permeants leaked through the interplaque regions if the particles alone limited transport. It has been shown that inflammation of the underlying muscle and lamina propria can disrupt the bladder permeability barrier by damaging tight junctions and apical membranes and causing sloughing of epithelial cells. Leakage of urinary constituents through the damaged epithelium may then exacerbate the inflammation in the deeper layers.
It was Parsons who hypothesized and popularized the concept that IC in a subset of patients is the result of some defect in the epithelial permeability barrier of the bladder surface glycosaminoglycans. The major classes of glycosaminoglycans (GAGs) include hyaluronic acid, heparin sulfate, heparin, chondroitin 4-sulfate and chondroitin 6-sulfate, dermatan sulfate, and keratan sulfate. These carbohydrate chains, coupled to protein cores, produce a diverse class of macromolecules, the proteoglycans. GAGs exist as a continuous layer on the bladder urothelium. Except heparin, all the other types of GAGs have been found on the bladder surface. The GAG layer functions as a permeability and antiadherence barrier. When impaired, its functions can be duplicated by exogenous GAG. In the absence of this protective layer in the urinary bladder, its susceptibility to infection would increase and the production of nitric oxide in the urothelial cells, and of substance P in the intraepithelial afferent C-fiber terminals, increases. Consequently, the permeability of both the urothelium and the blood vessels in the mucous membrane increases and the blood flow slows due to vasodilatation.
Parsons and Hurst reported a lower excretion of urinary uronic acid and glycosaminoglycans in IC patients than in normal volunteers and hypothesized that a leaky transitional epithelium might be absorbing these substances to its surface. The data are interesting in that one might expect urinary GAG to increase with injury to the bladder and decrease with resolution. The San Diego group went on to show experimentally that one can damage the GAG layer with protamine sulfate with resultant back-diffusion of urea through the bladder lumen and that this urea loss can be prevented with a bladder instillation of exogenous GAG (heparin). By placing a solution of concentrated urea into the bladder of IC patients and measuring absorption versus controls, Parsons and colleagues found support for their theory in patients with IC. The rationale of the epithelial permeability school is nicely summarized in four publications and provides a comprehensive, if somewhat imperfect, theory of the disorder.
Support for an epithelial abnormality from a different perspective has come from Bushman and associates, who found aneuploid DNA profiles on barbotage specimens from IC patients that may signal an underlying abnormality of the epithelial cell population in some patients with IC. Wilson and colleagues identified a loss of type IV collagen in the urothelial basement membrane in 5 of 11 IC patients. Hurst's group studied bladder biopsies of IC patients and controls and concluded that there is a deficit of bladder luminal and basal proteoglycans associated with the disorder. The basal abnormality may reflect an altered urothelial differentiation program. In a later study, IC bladder biopsies showed abnormalities in 24 of 27 patients when examined by immunohistochemical assessment of E-cadherin, ZO-1, uroplakin, and chondroitin sulfate. Erickson and coworkers measured a glycoprotein (epitectin) in the urine of IC patients and found a decrease compared with a control population, although a significant overlap was detected. Buffington and Woodworth gave 6 IC patients and 6 controls oral fluorescein dye. The IC patients had higher levels of fluorescein in their plasma and lower urinary excretion of the dye, suggesting altered membrane permeability and increased fluorescein reabsorption in the bladder wall of IC patients. Erickson and colleagues compared urinary levels of hyaluronic acid in IC patients and control subjects, reporting higher urinary hyaluronic acid in the patient group, possibly accounted for by leakage of this GAG across the epithelium.
Further data for an abnormal surface mucin came from Moskowitz and colleagues, who studied biopsies from 23 IC patients with regard to the presence of a glycoprotein component of the surface mucin referred to as GP1 and compared the results to 11 normal controls. Qualitative GP1 changes in a majority of IC patients were identified. GP1 reactivity was noted in all control subjects but was absent in 35% of IC patients and diminished in 61%. This study may provide evidence of an abnormal bladder urothelium, but the effects of bladder distention in the IC group are unknown and may have contributed to the results. No pathologic controls were used, and no attempt was made to correlate GP1 reactivity with IC symptoms. Castration in female rabbits is associated with bladder mucosal changes resulting in increased mucosal permeability. Birder and colleagues have shown that feline IC results in increased baseline production of nitric oxide due to inducible nitric oxide synthase. These changes in transmitter release may have a role in altering mucosal barrier properties.
Purportedly strong evidence for a population with mucosal leak has been reported. Parsons placed water or 0.4 M potassium chloride (KCl) intravesically into normal volunteers and IC patients. Water did not provoke pain in either group, but KCl provoked the symptom in 4.5% of normal individuals and 70% of IC patients. Symptomatic responses were reduced in patients on heparinoid therapy. Similar findings occur in patients with radiation cystitis (100%), urinary infection (100%), detrusor instability (25%), "urethral syndrome" (55%), and greater than 80% of women with endometriosis, vulvodynia, and pelvic pain. Eighty-four percent of men with prostatitis also have a positive test. The poor specificity of the KCl test does not suggest that it is providing unequivocal evidence of a permeability dysfunction. Because it is known that the normal bladder epithelium can never be absolutely tight, and there is always some leak, however small, it is conceivable that the findings of pain with KCl are related to a hypersensitivity of the sensory nerves in this condition, rather than to pathologic epithelial permeability, at least in some patients. In fact, KCl administered intravesically to cats with feline IC seems to inhibit afferent firing of peripheral A fibers. Heightened sensitivity of afferent nerve fibers can explain KCl results without necessarily evoking increased permeability. Intravesical administration of KCl has since been proposed as a diagnostic test for IC (see later).
How central abnormal epithelial permeability is to IC is, however, by no means clear. Tamm-Horsfall protein (THP), a high-molecular-weight glycoprotein synthesized exclusively by the ascending loop of Henle and the distal tubule of the kidney, has been studied as a potential marker of urothelial permeability. Fowler provided graphic data that the urothelium might be leaky in IC. With immunohistochemical techniques his group assayed the bladder biopsies of 14 IC patients and 10 normal controls for intraurothelial THP to assess indirectly the in-vivo permeability of the urothelium. Eight pathologic control subjects were also assessed. Ten of 14 IC patients versus 1 of 18 control subjects demonstrated intraurothelial THP. Serum THP autoantibody is higher in PBS/IC patients versus controls. It is known that excretion rates of THP vary widely, even in repeat samples taken from the same individual. Subsequent studies in IC have failed to show differences in the presence of intraurothelial THP in the IC population versus controls and in antibody reactivity to THP. Bade and coworkers failed to find THP in the bladder tissue from 10 IC patients. Others have suggested that when THP is seen in bladder tissue, it is an incidental finding of no clinical significance. The finding of intraurothelial THP has not been shown to be a harbinger of IC or any other bladder disorder.
Finally, we must look at a body of literature that has failed to find GAG abnormality or hyperpermeability. Ultrastructural, biochemical, and functional studies of bladder GAG have not supported this theory. Nickel's group reported sophisticated electron micrography using a specific anti-mucus, antisera stabilization technique to study the ultrastructural morphologic appearance of the GAG. No significant difference in the morphologic appearance of the mucus or GAG layer was noted in IC versus control subjects. Urinary chondroitin sulfates, heparan sulfate, and total sulfated glycosaminoglycans normalized to creatinine are not altered in IC. Although an increased ratio of total GAGs to sulfated GAGs in IC may indicate an altered GAG layer, whether it reflects a cause or is a result of the primary pathologic process(es) is unknown. That leaves one to postulate an as yet unknown functional abnormality, rather than GAG deficiency, to account for any increase in permeability.
Chelsky and coworkers measured bladder permeability in IC using direct measurement by transvesical absorption of 99mTc-diethylenetriaminepentaacetic acid (DTPA). Whereas some IC patients had a more permeable bladder than others, the same was true for normal volunteers. Increased permeability in the IC group could not be demonstrated. However, three IC patients had marked absorption of DTPA and may represent a subpopulation of patients with increased epithelial permeability. Intravesical instillation of 10% and 20% ethanol in rabbits was reported to be a reliable quantitative measure of bladder hyperpermeability by the San Diego group and subsequently failed to demonstrate bladder permeability in humans with IC.
Neurobiology
Neuropeptides present in primary afferents and the dorsal horn of the spinal cord have an important role in the mediation of nociceptive input under normal conditions. Under pathologic conditions, such as chronic inflammation or after peripheral nerve injury, the production of peptides and peptide receptors is dramatically altered, leading to a number of functional consequences. Inflammatory painful stimuli, especially if repeated, can chronically alter innervation, central pain-processing mechanisms, and tissue responses. It has been known for some time that the sensory nervous system can generate some of the manifestations of inflammation. Activation of capsaicin-sensitive afferent neurons locally and centrally may be involved in stress-related pathologic changes in the rat bladder. Activation of sensory nerves, specifically pain fibers, is known to trigger neurogenic inflammation through release of neuropeptides such as substance P, neurokinin A, and calcitonin gene–related protein, and subsequent increase in vascular permeability, with leukocyte adhesion and tissue edema. The neuropeptide mediators have been shown to also cause degranulation of mast cells with release of additional potent mediators of inflammation and to lead to injury and increased permeability of epithelial surfaces. An increase in nerve fibers within the suburothelium and detrusor muscle in ulcerative IC has been noted. A correlation was found between the number of nerve fibers and numbers of mast cells as well as between the number of nerve fibers and the amount of histamine. Consolidating the leaky urothelium theory and mast cell activation, neurogenic inflammation is an attractive proposal for etiology and can readily accommodate infectious, immunologic, and autoimmunologic mechanisms as factors.
KEY POINT: ETIOLOGY (BLADDER GLYCOSAMINOGLYCAN LAYER AND EPITHELIAL PERMEABILITY) |
| Overall, it does seem that there is a population of IC patients with increased epithelial permeability. Increased mucosal permeability is nonspecific and a consequence of bladder inflammation and also occurs with cyclophosphamide-induced bladder injury, bacterial infection, and cystitis after intravesical challenge with antigen after sensitization. It may also be a consequence of aging itself. Whether this represents a primary cause of IC or merely reflects the result of an as-yet-unidentified source of inflammation is unclear. Treatments that tend to damage GAG, including transurethral resection and laser of ulcerated areas, bladder distention, silver nitrate administration, and oxychlorosene (Clorpactin) administration and use of the organic solvent dimethyl sulfoxide (DMSO) have all been used with varying results to treat IC. Increased permeability and epithelial dysfunction must be only a part of the story. |
Harrison and associates proposed that small-diameter sensory nerves in the bladder wall may have a role in the transmission of the sensation of pain and in the triggering of inflammatory reactions rather than forming the afferent limb of the micturition reflex. Abelli and colleagues demonstrated in the rat urethra that mechanical irritation alone can cause neuropeptide release from peripheral capsaicin-sensitive primary afferent neurons resulting in neurogenic inflammation. Extracellular adenosine triphosphate (ATP) can act through the purinergic receptor subtype P2X3 to transmit a pain signal to the central nervous system. These subunits expressed by cultured IC bladder urothelial cells are upregulated during in-vitro stretch and may phenotypically mimic sensory neurons.
Several pieces of additional information support a theory of neurogenic inflammation. Levels of nerve growth factor are elevated in bladder biopsy specimens of IC patients. Studies in rats using pseudorabies virus clearly show that bladder inflammation can be induced from a somatic structure through a neural mechanism and that central nervous system dysfunction can bring about a peripheral inflammation. Pelvic nerve stimulation in the rat increases urothelial permeability that is antagonized by capsaicin, indicating both an efferent effect of afferent nerves and afferent mediated neuroepithelial interaction.
Numerous studies indicate increased sympathetic activity in IC. Hohenfellner and colleagues suggested that IC is associated with increased sympathetic outflow into the bladder and altered metabolism of vasoactive intestinal polypeptide and neuropeptide Y. Neuropeptide Y inhibits bladder afferents and therefore may be involved in autonomic disturbances affecting the bladder. Elevation of urinary catecholamines in IC patients and of plasma catecholamines in cats with feline IC has been observed, as has an increased density and number of nerve fibers immunoreactive for tyrosine hydroxylase in IC patients. Whether these changes reflect a cause of IC or are merely the result of long-standing intense pain and a severely pathologic voiding pattern is unknown.
Galloway and colleagues proposed that the changes in IC may be explained by an increase in sympathetic discharge, analogous to that seen in reflex sympathetic dystrophy (RSD) of limbs. The pathology in RSD is the development of abnormal synaptic activity between sensory afferent and sympathetic efferent neurons. Nerve cells in the spinal cord become hypersensitive to sensory input, and this sustains abnormal sympathetic outflow and corresponding vasomotor dysregulation. The excess sympathetic outflow leads to constriction of blood vessels and tissue ischemia, setting up further sensory changes and perpetuating the cycle. In RSD, there is usually a trigger event leading to these changes. With the acute phase of RSD, regional signs of inflammation are evident in the affected extremity. One school of thought believes an inflammatory response to an injury initiates RSD. Increased capillary permeability is a direct result. Perhaps a urinary infection could trigger such a pathologic cycle in some IC patients?
Herbst and colleagues produced bladder lesions in a dog resembling the ulceration of IC by ligating the blood vessels to the posterior bladder wall and infecting the area with Streptococcus viridans. Studies using laser Doppler flowmetry have shown that when the bladder is distended under anesthesia, blood flow increases in control patients to a statistically significant degree as compared with IC patients. Another study has purported to show that topical heparin therapy can normalize urothelial permeability and vesical blood flow in IC. Decreased microvascular density has been described in the suburothelium but not in the deeper mucosa in bladder biopsies of women with IC. Hyperbaric oxygen has been suggested to be effective in the treatment of PBS/IC as well as radiation-induced cystitis.
If lumbar sympathetic blocks can decrease the pain of IC, a role of the sympathetic nervous system in IC pathogenesis is a reasonable supposition. An increase in sympathetic activity has been demonstrated in cats with feline IC. Similar findings have been reported in a small study of IC patients and sympathetic activity may be an underlying common denominator in many disorders associated with PBS/IC.
Nevertheless, no studies performed to date can say any case of IC is related to the syndrome of RSD. No single test can be used to exclude sympathetically maintained pain, and there are no clear symptoms that predict sympathetically mediated pain. In the animal model, bladder ischemia is associated with detrusor overactivity or impaired detrusor contraction, not sensory urgency. Those patients with RSD who have voiding symptoms rarely have a picture that would be confused with IC.
Before leaving the neurogenic theory of etiology, it is important to note that the nervous system itself almost surely contributes to the chronic nature of this pain syndrome, regardless of initiating etiology. Repetitious stimulation of a peripheral nerve, at sufficient intensity to activate C fibers, results in a progressive buildup of the magnitude of the electrical response recorded in the second-order dorsal horn neurons. This "wind-up" phenomenon is central to the concept of chronic pain. Biochemically it is dependent on activation of N-methyl-d-aspartate (NMDA) receptors in the spinal cord. With persistent NMDA receptor activation, spinal cord cells undergo trophic changes, and the pain resulting from subsequent stimulation becomes exaggerated and prolonged. This "pain memory" in the spinal cord may be what causes IC patients to become refractory to different therapies. NMDA-receptor-driven formation of new connections in the spinal cord may account for the expansion of the pain field.
Upregulation of the CNS and augmented sensory processing has been referred to as non-nociceptive pain. The four characteristic features of non-nociceptive pain would seem to apply very well to the clinical syndrome of IC. Chronic neuropathic pain may continue after the resolution of tissue damaablge and persist on the basis of a maladaptive mechanism.
| Table 3. Non-nociceptive Pain: Characteristic Clinical Features. |
|
Burnstock's observation that ATP has a role in mechanosensory transduction by the epithelial lining of hollow viscus organs such as bladder has been followed up by Sun and colleagues. Stretched epithelial cells lining hollow organs release ATP, which acts on purinergic nociceptive receptors on subepithelial sensory nerve terminals. ATP was significantly elevated in the urine of PBS/IC patients, and the stretch-activated release of ATP was augmented in IC urothelium.
KEY POINT: ETIOLOGY (NEUROBIOLOGY) |
| Neurogenic inflammation may be the cause of some cases of IC or may be the result of other initiating etiologic events. It is not incompatible with the central role of the mast cell, or with the leaky epithelium theory. It conceivably could result in the appearance of autoimmune phenomena or result from an episode of infection. The central nervous system may also be implicated in dysregulation of the pelvic floor, resulting in chronic pelvic pain and contributing to IC, and perhaps in the rare cases of IC that chronologically seem to relate to trauma or pelvic surgery. It is an etiologic theory that provides fertile ground for new treatment possibilities. |
Urine Abnormalities
Current theories of pathogenesis generally involve access of a component of urine to the interstices of the bladder wall, resulting in an inflammatory response induced by toxic, allergic, or immunologic means. The substance in the urine may be a naturally occurring one — a substance that acts as an initiator only in particularly susceptible individuals — or may act like a true toxin, gaining access to the urine by a variety of mechanisms or metabolic pathways. Clemmensen and coworkers noted that 8 of 11 IC patients had positive skin reactions to patch tests with their own urine. Immediate reactions were not observed, and the histology suggested a toxic rather than an allergic reaction. Lynes and associates were unable to find a urinary myotropic substance unique to IC patients. The San Diego group found IC urine to result in higher cell death of cultured transitional cells than normal urine, suggesting a toxic compound in the urine of some IC patients . They identified heat-labile, cationic components of low molecular weight that bind to heparin and that when separated from the bulk of urinary wastes are cytotoxic to urothelial cells as well as underlying smooth muscle cells. They reported a 12% increase in 72-kD stress protein in cells treated with urine from IC patients compared with controls.
Others have not been able to demonstrate in-vitro cytotoxicity or immunohistochemical changes in the nociceptive centers in the spinal cord or bladder wall when IC urine was compared with control urine. Efforts to induce an IC-like picture in the rabbit bladder from exposure to urine of IC patients have failed to demonstrate conclusive changes. Increased levels of soluble mediators associated with activation of sensory neurons and/or mast cells have been found in the urine of both IC and bladder cancer patients.
Circumstantial evidence for the toxicity of IC urine is suggested by the failure of substitution cystoplasty and continent diversions in some of these patients because of the development of pain or contraction of the bowel segment over time and the histologic findings similar to IC found to occur in bowel used to augment the small-capacity IC bladder. Intestinal mucosa in contact with urine undergoes progressive changes as long as 3 years after surgery and the significance of the histologic IC-like changes has been questioned.
Antiproliferative Factor (APF)
The finding that cells from the bladder lining of normal controls grow significantly more rapidly in culture than cells from IC patients led Keay and associatesat the University of Maryland to the discovery of an antiproliferative factor (APF) produced by the urothelium of IC patients. Normal bladder cells were cultured in the presence of urine from patients with IC, asymptomatic controls, bacterial cystitis, and vulvovaginitis. Only urine from IC patients inhibited bladder cell proliferation. The presence of APF was found to be a sensitive and specific biomarker for IC (Table 4). It was found in bladder urine but not in renal pelvic urine of IC patients, indicating production by the bladder urothelial cells. Subsequent studies indicated that APF is associated with decreased production of heparin-binding epidermal growth factor–like growth factor (HB-EGF). APF activity was related to increased production of epidermal growth factor (EGF), insulin-like growth factor-1, and insulin-like growth factor–binding protein-3 by the bladder cells from IC patients but not by the cells from healthy bladders. Studies of IC patients and asymptomatic controls showed urine levels of APF, HB-EGF, and EGF to reliably separate out IC from controls.
| Table 4. Prevalence of Urine Antiproliferative Factor Activity in Interstitial Cystitis Patients and Control Groups. | ||
Groups |
No. of Patients (Positive/Total) |
% Positive |
Patients |
||
Interstitial cystitis |
206/219 |
94 |
Controls |
||
Asymptomatic |
10/113 |
9 |
Overactive bladder |
2/32 |
6 |
Bacterial cystitis |
7/58 |
12 |
Microscopic hematuria |
2/19 |
10 |
Stress incontinence |
1/10 |
10 |
Neurogenic bladder |
0/11 |
0 |
Benign prostatic hypertrophy |
1/14 |
7 |
Nonbacterial prostatitis |
1/16 |
6 |
Vulvovaginitis |
0/12 |
0 |
Miscellaneous |
1/16 |
6 |
APF levels in the urine were found to discriminate between men with IC versus those with chronic pelvic pain syndrome (CPPS) or nonbacterial prostatitis. APF activity dropped significantly in IC patients within 2 hours after hydrodistention and after 5 days of sacral neuromodulation. Cell culture studies showed that APF actually caused decrease in HB-EGF and increase in EGF, mirroring the differences in urine levels of these growth factors between IC patients and controls and suggesting that APF is the primary abnormality.
Whereas APF may prove to be a useful marker for PBS/IC, it may also unlock the etiology of the syndrome. It has been hypothesized by Keay and colleaguesthat PBS/IC may result from an inhibition of bladder epithelial cell proliferation caused by the APF, which is mediated by its regulation of growth factor production from bladder cells. Conceivably, any of a variety of injuries to the bladder (infection, trauma, and overdistention) in a susceptible individual may result in PBS/IC if APF is present and suppresses production of HB-EGF. Theoretically, if production of APF could be "turned off" by genetic techniques, or its effects were nullified by exogenous HB-EGF growth factor, the clinical syndrome might be prevented.
APF has been purified (Fig. 6) and proved to be a frizzled 8 protein that belongs to a newly discovered family of proteins that seem to be important in the development of nerve tissues, skin, and the lining of organs. Studies are ongoing to confirm the research by Dr. Keay and colleagues and expand on its significance in diagnosis and development of a rational treatment approach.
|
| Figure 6. Composition and structure of antiproliferative factor (APF). |
KEY POINT: ETIOLOGY (ANTIPROLIFERATIVE FACTOR) |
| Antiproliferative factor is a frizzled 8 protein produced by bladder uroepithelial cells of PBS/IC patients. It inhibits bladder cell proliferation and appears to be a sensitive and specific biomarker for IC. Initial studies suggest it can differentiate chronic pelvic pain syndrome in men/chronic nonbacterial prostatitis from PBS/IC. It causes a decrease in heparin binding epidermal growth factor-like growth factor and an increase in epidermal growth factor. It may ultimately unlock the etiology of the syndrome and could provide avenues for development of future therapies. |
Other Potential Causes
Various other etiologic theories have been proposed, but none has received much scientific support. Voiding almost hourly, always having to be aware of how far the nearest restroom facilities are, and suffering constant pain would be expected to lead to psychological stress. However, could there be individual differences in the propensity to develop IC that result from a dysregulation of anxiety and mood? There are no data currently to suggest that stress initiates the chronic syndrome of IC, although it certainly can increase symptom severity. Cats restricted to indoor living are five times more likely to have urinary problems as cats allowed outdoors. Female patients with PBS/IC have been shown to have increased heart rate at baseline and throughout a laboratory mental stress challenge but did not demonstrate greater autonomic reactivity to stress. Until stress can be shown to produce PBS/IC de novo in humans, it is just as reasonable to speculate that the stress is a result of the syndrome as a primary cause for it.
Speculation that abnormalities in or obstruction of lymphatics or vascular structures is causative has never been borne out. The fact that some of these patients have had hysterectomy probably relates more to the attempt of the physician to treat chronic pelvic pain than postsurgical change causing the IC syndrome.
The knowledge that there is at least a 5:1 female to male preponderance immediately makes the role of the hormonal milieu potentially important. Paradoxically, it is known that estrogens can control hematuria in hemorrhagic cystitis, perhaps by decreasing the fragility of the mucosal microvasculature of the bladder. Estradiol augments while the estrogen receptor blocker tamoxifen inhibits mast cell secretion. Bladder mast cells express high-affinity estrogen receptors, and there is a higher number of such cells present in patients with IC compared with controls. Although this may help explain why IC is so common in women, the hormonal role can only account for the propensity of IC to occur in females, not the ultimate etiology.
Pelvic floor dysfunction has been associated with PBS/IC for many years, and uncontrolled trials suggest that treatment of the pelvic floor can be effective in ameliorating symptoms. Speculation that abnormalities of the pelvic floor muscular function may contribute to the etiology of some cases of the chronic pelvic pain syndrome in men are well accepted, and a similar case might be made for patients with PBS/IC, although scientific support for a direct etiologic relationship is lacking.
PATHOLOGY
One can have pathology consistent with the diagnosis of IC, but there is no microscopic picture pathognomonic of this syndrome. The role of histopathology in the diagnosis of IC is primarily one of excluding other possible diagnoses. One must rule out carcinoma and carcinoma in-situ, eosinophilic cystitis, tuberculous cystitis, as well as any other entities with a specific tissue diagnosis.
|
| Figure 7. Knifelike Hunner's ulcer in interstitial cystitis. (H&E, original magnification, x40.) |
 |
| Figure 8. Nonulcerative form of interstitial cystitis with dense lymphoid infiltrate in the lamina propria. (H&E, original magnification, x20.) |
Although earlier reports described a chronic, edematous pancystitis with mast cell infiltration, submucosal ulcerations and involvement of the bladder wall, and chronic lymphocytic infiltrate, these were cases culled from patients with severe disease and not representative of the majority of cases currently diagnosed. The pathologic findings in IC are not consistent. There has been a great variation in the reported histologic appearance of biopsy specimens from IC patients, and even variation among specimens taken from the same patients over time.
Lepinard and colleagues reported a pancystitis affecting the three layers of the bladder wall. In nonulcerative disease the vesical wall was never normal, with epithelium being thinned and muscle being affected. Johansson and Fall looked at 64 patients with ulcerative disease and 44 with nonulcerative IC. The former group had mucosal ulceration and hemorrhage, granulation tissue, intense inflammatory infiltrate, elevated mast cell counts, and perineural infiltrates. The nonulcer group, despite the same severe symptoms, had a relatively unaltered mucosa with a sparse inflammatory response, the main feature being multiple, small, mucosal ruptures and suburothelial hemorrhages that were noted in a high proportion of patients. Because these specimens were almost all taken immediately after hydrodistention, how much of the admittedly minimal findings in the nonulcer group were purely iatrogenic is a matter of speculation. One can see completely normal biopsies in the nonulcerative IC group. Transition from nonulcerative to ulcerative IC is a rare event, and pathologically the two types of IC may be completely separate entities. Whereas mast cells are more commonly seen in the detrusor in ulcerative IC, they are also common in patients with idiopathic bladder instability. Mastocytosis in PBS/IC is best documented by tryptase immunocytochemical staining. Despite attempts to develop a diagnostic algorithm based on the detrusor to mucosa mast cell ratio and nerve fiber proliferation, mast cell counts per se have no place in the differential diagnosis of this clinical syndrome.
Lynes and coworkers concluded that biopsy specimens are often not helpful in confirming the diagnosis. Although IC patients in his study had a higher incidence and degree of denuded epithelium, ulceration, and submucosal inflammation, none of these findings was pathognomonic. In addition, these "typical" findings occurred only in IC patients with pyuria or small bladder capacity. Epithelial and basement membrane thickness, submucosal edema, vascular ectasia, fibrosis, and detrusor muscle inflammation and fibrosis were not significantly different in the IC and control patients.
Attempts to definitively diagnose IC by electron microscopy have also been very unsuccessful. Collan's group, in the first such study, wrote that the similarity of the ultrastructure of epithelial cells in controls and IC patients makes it improbable that the disease process originates in the epithelium. Other investigators found no differences in the morphologic appearances of the glycocalyx and of urothelial cells in patients with IC when compared with controls. Anderstrom and colleagues saw no surface characteristics specific for IC but believed that the mucin layer covering the urothelial cells seemed reduced in IC compared with controls, a fact disputed in a very elegant paper. Elbadawi and Light observed ultrastructural changes sufficiently distinctive to be diagnostic in specimens submitted for pathologic confirmation of nonulcerative IC. Marked edema of various tissue elements and cells appeared to be a common denominator of many observed changes. The wide-ranging discussion of the etiology of IC in Elbadawi's papers is fascinating, but the pathologic findings are potentially marred by the methodology, in that specimens were obtained after diagnostic hydrodistention.
So what is the place of pathologic examination of tissue in IC? Attempts to classify the painful bladder by the pathoanatomic criteria described by Holm-Bentzen are of questionable value. There is a group of patients with what she and her colleagues describe as "nonobstructive detrusor myopathy". In this series these patients with degenerative changes in the detrusor muscle often had residual urine, a history of urinary retention, and an absence of sensory urgency on cystometry with bladder capacities over 400 mL. Most would not be clinically confused with IC. A similar English series, however, included patients who met NIDDK research criteria and associated detrusor myopathy with diminished detrusor compliance and ultimate bladder contracture.
The ICDB study worked backward from symptoms to pathology and concluded that certain symptoms are predictive of specific pathologic findings (Table 5). Denson and coworkers analyzed forceps biopsies from 65 females and 4 males with PBS/IC. Ten percent of specimens showed vasodilatation or submucosal edema. Inflammation was absent in 30% of patients and mild in another 41%. Cystoscopic changes did not correlate with degree of inflammation. Hanus and colleagues studied 84 biopsy specimens from 112 PBS/IC patients and reported a linear relationship between the mean bladder capacity under anesthesia and severity of glomerulations. They did not find a correlation between severity of symptoms and histopathologic changes observed by light or electron microscopy.
| Table 5. Associations among Pathologic Features and Patient Symptoms. |
Night-Time Frequency
Urinary Urgency
Urinary Pain
|
Rosamilia and colleagues reviewed the pathology literature pertaining to PBS/IC in two recent publications and presented their own data. They compared forceps biopsies from 35 control and 34 PBS/IC patients, 6 with bladder capacities less than 400 mL under anesthesia. Epithelial denudation, submucosal edema, congestion and ectasia, and inflammatory infiltrate were increased in the PBS/IC group. Submucosal hemorrhage did not differentiate the groups, but denuded epithelium was unique to the IC group and more common in those with severe disease. The most remarkable finding in this study was that histologic parameters were normal and indistinguishable from control subjects in 55% of IC subjects. Method of biopsy can be important in interpreting findings, because transurethral resection biopsies tend to show mucosal ruptures, submucosal hemorrhage, and mild inflammation while histology is normal approximately half the time with cold-cup forceps biopsie.
KEY POINT: PATHOLOGY |
| Histopathology plays a supportive diagnostic role at most. While recent studies suggest that a severely abnormal pathology may be associated with poor prognosis, this is not necessarily the case. IC is a diagnosis of exclusion, and, at this point in time, excluding other diseases that are pathologically identifiable is the primary utility of bladder biopsy in this group of patients. |
DIAGNOSIS
PBS/IC can be considered one of the chronic visceral pain syndromes, affecting the urogenital and rectal area, many of which are well described but poorly understood. These include vulvodynia, orchialgia, penile pain, perineal pain, and rectal pain. In men, many of the entities have now been folded into the rubric of the chronic pelvic pain syndrome and can be difficult to distinguish from PBS/IC. The diagnosis of PBS/IC is by its very nature based on the definition. In the past this was by default, the symptom criteria enumerated by the NIDDK (see Table 1). It has now morphed largely into a diagnosis of chronic pain, pressure, or discomfort associated with the bladder, usually accompanied by urinary frequency in the absence of any identifiable cause. Diagnostic approaches vary widely, and general agreement on a diagnostic algorithm remains a future goal. The disorder can be very difficult to diagnose until symptoms become well established, unless one has a high level of suspicion. Frequency and pelvic pain of long duration related to the bladder unrelated to other known causes establishes a working diagnosis. It is often difficult for patients to distinguish between sensations of pain, pressure, discomfort, and urgency. Ask a patient why he or she voids hourly, and it is usually because of discomfort rather than convenience. Heavy reliance on other aspects of the NIDDK research criteria will result in underdiagnosing more than half of patients. IC symptom scales, like the AUA symptom score for benign prostatic hyperplasia, are designed to evaluate the severity of symptomatology and monitor disease progression or regression with or without treatment. They have not been validated as diagnostic criteria.
One must rule out infection and less common conditions, including but not limited to carcinoma, eosinophilic cystitis, malacoplakia, schistosomiasis, scleroderma, and detrusor endometriosi. In men younger than the age of 50, video-urodynamics are useful to rule out voiding dysfunction resulting from vesical neck obstruction, "pseudo" dyssynergia, or impaired contractility. Musculoskeletal dysfunction may also play a role in causation or increasing symptom severity and should be looked for in the diagnostic phase of evaluation. Reports of successful treatment of IC symptoms by laparoscopic adhesiolysis or urethral diverticulum excision give credence to the fact that IC is a diagnosis of exclusion. Many drugs including cyclophosphamides, aspirin, nonsteroidal anti-inflammatory agents, and allopurinol have caused a nonbacterial cystitis that resolves with drug withdrawal.
Various gynecologic problems can mimic the pain of IC. The pelvic congestion syndrome, a condition of the reproductive years and equally prevalent among parous and nulliparous women, is manifest by shifting location of pain, deep dyspareunia and postcoital pain, and exacerbation of pain after prolonged standing. Similar symptoms can be seen in PBS/IC. Other gynecologic disorders can include pelvic tumors, vaginal atrophy, vulvodynia, vestibulitis, pelvic relaxation, pelvic adhesive disease, levator ani myalgia, and undiagnosed chronic pelvic pain. Endometriosis probably causes pain, but that conclusion has to be regarded carefully, because it is based largely on one study. Whiteside and Falcone point out that any claim linking endometriosis with pain fails to account for the common experience that identical lesions can be found in symptomatic and asymptomatic women. Between 2% and 43% of asymptomatic women are found to have endometriosis. Furthermore, there does not appear to be any risk for patients with asymptomatic mild endometriosis to develop symptoms even after greater than 10 years. While 70% to 90% of women with chronic pelvic pain have endometriosis, this does not definitively establish causation. Laparoscopy, which is not considered essential before initiating hormonal treatment of endometriosis, should not be considered a part of any routine evaluation of PBS/IC unless a gynecologist believes it is likely to benefit the patient.
A presumptive diagnosis can be made merely by ruling out known causes of frequency and pain/urgency in a patient with compatible chronic symptoms. Often this will involve a complete history, physical examination, appropriate cultures, and local cystoscopy. In the absence of microhematuria, the value of cytology is questionable but something we still consider important, especially if bladder carcinoma in situ is a serious possibility, as in patients older than 40 and those with a smoking history. The recent report of a large series of PBS/IC patients indicating that 1% actually had transitional cell carcinoma and that 4 of the 6 cancer patients did not have microhematuria provides evidence for the justification of local cystoscopic examination.
It must be recognized that one may sacrifice a certain level of confidence in the diagnosis without the supporting evidence that can be furnished by additional studies. In a long-term illness such as IC, many patients and physicians ultimately want to base a diagnosis and treatment plan on the most complete data set possible. A more thorough evaluation for IC would also include a urodynamic evaluation and cystoscopy under anesthesia with hydrodistention of the bladder. Bladder biopsy is indicated only if necessary to rule out other disorders that might be suggested by the cystoscopic appearance. Cystoscopy under anesthesia with bladder distention has been important in the identification of Hunner's ulcer. Experimental data suggest that measurement of increased nitric oxide levels in the bladder can also accurately identify those with ulcerative disease. The diagnosis is generally subject to more rigorous testing in Europe than in North America, where symptoms in the absence of other obvious causes seems to be the gold standard.
Although sensations reported during cystometric bladder filling are subjective, they have a normal pattern and may be helpful in distinguishing bladder pathology. Many dispute the need for urodynamic study, but we agree with Siroky that not only can it help to assess bladder compliance and sensation and reproduce the patient's symptoms during bladder filling, but it can also help to rule out detrusor overactivity. Women with pain on bladder filling can be indistinguishable from those with detrusor overactivity in their perception of bladder fullness. One should be wary of diagnosing IC in patients with discrete, involuntary bladder contractions whose symptoms respond to anticholinergic therapy. The two problems coexist in 15% to 19% of patients, but the pathophysiology is likely to be very different. Patients who respond to anticholinergic medication tend not to respond to standard therapy for IC. If involuntary contractions are noted and the patient's symptoms of frequency and pain continue despite successful treatment, one is on firmer ground in considering a diagnosis of IC. Complex cases may benefit from full video-urodynamics studies.
Cystometry in conscious IC patients generally demonstrates normal function, the exception being decreased bladder capacity and hypersensitivity. Pain on bladder filling that reproduces the patient's symptoms is very suggestive of IC. Bladder compliance in patients with IC is normal, because hypersensitivity would prevent the bladder from filling to the point of noncompliance. The possible addition of including a second cystometrogram after instillation of intravesical lidocaine to help to determine if pain is bladder related is a provocative one worth further study.
Long before it was considered a diagnostic tool, cystoscopy with hydrodistention of the bladder was used as a therapeutic modality for IC. Performing this procedure with the patient under anesthesia allows for sufficient distention of the bladder to afford visualization of either glomerulations or Hunner's ulcers (Figs. 9 and 10). After filling to 80 cm H2O for 1 to 2 minutes, the bladder is drained and refilled. The terminal portion of the effluent is often blood tinged. Reinspection will reveal the pinpoint petechial hemorrhages that develop throughout the bladder after distention and are not usually seen after examination without anesthesia.
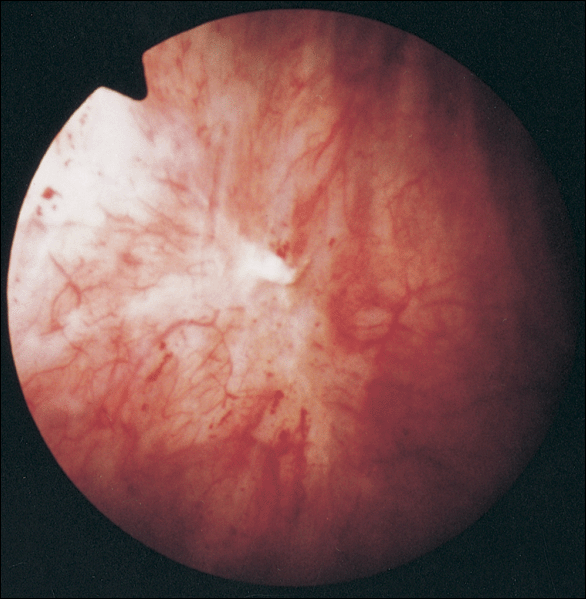
|
| Figure 9. Typical appearance of Hunner's ulcer in a patient with interstitial cystitis before bladder distention. |
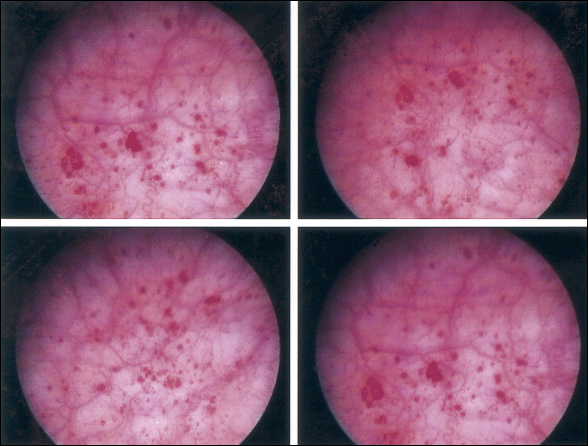
|
| Figure 10. Typical appearance of glomerulations after bladder distention in a patient with nonulcerative interstitial cystitis. |
Glomerulations are not specific for IC, and only when seen in conjunction with the clinical criteria of pain and frequency can the finding of glomerulations be viewed as significant. Glomerulations can be seen after radiation therapy, in patients with carcinoma, after exposure to toxic chemicals or chemotherapeutic agents, and often in patients on dialysis or after urinary diversion when the bladder has not filled for prolonged periods. They have been reported in the majority of men with prostate pain syndromes, begging the question as to whether the chronic pelvic pain syndrome in men is closely linked with IC. We have speculated that they may simply reflect the response of the bladder to distention after a prolonged period of chronic underfilling because of sensory urgency, rather than result from a primary pathologic process. While the presence of a Hunner's ulcer has been associated with pain and urinary urgency, neither the findings of bloody irrigating fluid nor glomerulations are strongly associated with any particular symptom in patients in the ICDB.
Further confusion arises when the patient demonstrates the symptoms of IC but the cystoscopic findings under anesthesia are completely normal. This occurred in 8.7% of patients undergoing cystoscopy with hydrodistention entered into the ICDB. Awad and colleagues recognized this entity soon after the NIDDK research criteria had been described. They reported on a series of patients in whom the symptomatology, urodynamic evaluation, histology, and response to therapy was identical to IC, in whom findings on cystoscopy with hydrodistention were normal. It was referred to as "idiopathic reduced bladder storage". Clinical, urodynamic, and cystoscopic data strongly suggest that the presence of glomerulations is not selecting out a meaningful difference in patients with symptoms of PBS/IC. The presence of glomerulations on cystoscopy under anesthesia meeting the NIDDK criteria may identify a group of patients with worse daytime frequency and nocturia and lower bladder capacity under anesthesia but does not have any relationship to biopsy findings, bladder pain, or urgency. Ultimately, disease definitions are helpful only relative to the utility they provide in terms of treatment and prognosis, and it is likely that the definition of IC and therefore proper methods of diagnosis will continue to evolve.
Markers
What is the value of a "diagnostic test" in what is essentially a clinical syndrome defined by a symptom-complex? If a patient has chronic pain associated with the bladder usually accompanied by urinary frequency with no discernible cause, we diagnose PBS/IC. In essence, once we have ruled out well-characterized pathologic entities, the patient makes the diagnosis by relating symptoms; much like a patient with impotence makes that diagnosis. Testing for impotence may give clues as to etiology, but we cannot rule out impotence in a patient who cannot function sexually by doing a test!
This is not to say that establishment of a valid diagnostic marker would not be a major advance in our understanding of IC. It will be important largely to the extent that it can predict prognosis in a given group of patients, predict response to therapy in a given group of patients, and/or distinguish between IC and another possible cause of the symptom-complex that has been diagnosed. Ultimately, marker identification may enable us to stratify patients with the symptom-complex in such a way that treatments will be specific to the specific etiology (disease) the patient has. Because various causes are identifiable, the diagnosis of IC may itself become a rarity, much as what has happened to "acute urethral syndrome".
In just such an effort, numerous investigators have looked at the mast cell as a possible diagnostic marker for IC. The results have been very contradictory, and at this time, in terms of the use of mast cell criteria in diagnosis, the issue remains moot. Methylhistamine, a histamine metabolite found in the urine and thought to reflect mast cell activation, was not associated with symptom scores, response to bladder distention, cystoscopic findings, or bladder biopsy features including mast cell determination by tryptase staining.
Attempts have been made to look at other markers, including eosinophil cationic protein, glycosaminoglycan excretion, and urinary histamine and methylhistamine. Proposals for measuring smooth muscle isoactin expression and urinary levels of neurotrophin-3, nerve growth factor, glial cell line–derived neurotrophic factor, and tryptase have been suggested. Low levels of GP51, a urinary glycoprotein with a molecular weight of 5 kD have been documented in IC patients compared with normal controls and patients with other urinary tract diseases. Even cell cultures and the measurement of elevated nitric oxide levels in air instilled and incubated in the bladder have been proposed for office screening.
The urine antiproliferative factor (APF) identified by Keay (see earlier) may prove to be the most accurate marker of PBS/IC when confirmed by other centers. It appears to have the highest sensitivity and specificity of the variety of possible markers tested and fits nicely into an etiologic schema. It has also been shown to differentiate men with PBS/IC symptoms from controls and differentiate men with bladder-associated pain and irritative voiding symptoms from those with pelvic/perineal pain alone and other nonspecific findings compatible with the chronic pelvic pain syndrome in men, previously referred to as "nonbacterial prostatitis". This evidence that chronic pelvic pain syndrome and PBS/IC are very likely two different disorders will doubtless be the subject of future research.
Potassium Chloride Test
Parsons has championed an intravesical KCl challenge, comparing the sensory nerve provocative ability of sodium versus potassium using a 0.4 M KCl solution. Pain and provocation of symptoms constitutes a positive test. Whether the results indicate abnormal epithelial permeability in the subgroup of positive patients or hyper-sensitivity of the sensory nerves is unclear. Normal bladder epithelium can never be absolutely tight, and there is always some leak, however small. The concentration of potassium used is 400 mEq/L, far exceeding the physiologic urinary concentrations of 20 to 80 mEq/L, depending on dietary intake. Healthy control subjects can distinguish KCl from sodium chloride, although they do not experience severe pain. The hope is that this test may stratify patients into those who will respond to certain treatments (perhaps those designed to fortify the glycosaminoglycan layer), but to date this information is lacking.
Used as a diagnostic test for IC, the KCl test is not valid. The gold standard in defining PBS/IC for research purposes has been the NIDDK criteria. These criteria are recognized to constitute a set of patients that virtually all researchers can agree have PBS/IC, although they are far too restrictive to be used in clinical practice. Thus, this group of patients should virtually all be positive if the KCl test is to have the sensitivity needed to aid in diagnosis. Up to 25% of patients meeting the NIDDK criteria will have a negative KCl test. In the group it should perform the best in, it is lacking in sensitivity. When we look at the specificity side of the equation, in the universe of asymptomatic persons, it performs relatively well and is rarely positive, although a recent study reported a 36% false-positive rate in asymptomatic men. It is in the patient population with confounding conditions for which we would want help in sorting out PBS/IC from other disorders. Twenty-five percent of patients with overactive bladder test positive, and virtually all patients with irritative symptoms from radiation cystitis and urinary tract infection test positive. The results with chronic prostatitis/chronic pelvic pain syndrome in men are variable, but 50% to 84% of men have been reported to test positive. In women with pelvic pain, results are similar; and based on these findings, Parsons and colleagues have expressed the view that PBS/IC may affect over 20% of the female population of the United States. Another way to interpret the findings would be that the KCl test is very nonspecific, missing a significant number of PBS/IC patients and overdiagnosing much of the population.
Cystometry employing KCl (0.3 M) has been compared with saline cystometry in patients with PBS/IC and those with detrusor overactivity. Both groups show lowered volumes at first desire to void and lower cystometric capacity with KCl, raising the question as to whether potassium may act not on the urothelial sensory mechanism but rather on the detrusor muscle.
Prospective and retrospective studies looking at the KCl test for diagnosis in patients presenting with symptoms of PBS/IC have found no benefit of the test in comparison with standard techniques of diagnosis. The development of a painless modification of the KCl test using cystometric capacity and a 0.2 M solution may improve acceptability among patients, but further research is needed to determine what place, if any, this test will have in the diagnostic or treatment algorithm for PBS/IC.
CLINICAL SYMPTOM SCALES
There are three published PBS/IC symptom questionnaires: the University of Wisconsin IC Scale, the O'Leary-Sant IC Symptom Index and IC Problem Index, and the Pelvic Pain and Urgency/Frequency (PUF) Scale.
The University of Wisconsin IC Scale (Table 6) includes 7 PBS/IC symptom items (1, 2, 10, 18, 21, 23, and 25) but has not been validated for identification or diagnosis of PBS/IC. It captures severity of symptom expression. PBS/IC patients do not appear to indiscriminately report higher scores than controls for different somatic and general complaints. Unlike the other two instruments, it addresses some quality-of-life issues, and this is an advantage when such issues are subjects of investigation. Its most attractive aspects are its clinically apparent face validity and its ease of implementation.
| Table 6. University of Wisconsin Symptom Instrument. | |
Symptom |
Score 1 to 6 (0 = Not at all; 6 = A lot) |
| 1. Bladder discomfort | |
| 2. Bladder pain | |
| 3. Other pelvic discomfort | |
| 4. Headache | |
| 5. Backache | |
| 6. Dizziness | |
| 7. Feelings of suffocation | |
| 8. Chest pain | |
| 9. Ringing in ears | |
| 10. Getting up at night to go to the bathroom | |
| 11. Aches in joints | |
| 12. Swollen ankles | |
| 13. Nasal congestion | |
| 14. Flu | |
| 15. Abdominal cramps | |
| 16. Numbness or tingling in fingers or toes | |
| 17. Nausea | |
| 18. Going to the bathroom frequently during the day | |
| 19. Blind spots or blurred vision | |
| 20. Heart pounding | |
| 21. Difficulty sleeping because of bladder symptoms | |
| 22. Sore throat | |
| 23. Urgency to urinate | |
| 24. Coughing | |
| 25. Burning sensation in bladder | |
The O'Leary-Sant indices (Table 7) form a validated questionnaire that was originally developed by focus groups, subjected to test-retest reliability analysis, and validated by administration to IC patients and asymptomatic controls. The questionnaires center on three questions related to urgency/frequency and one on bladder-associated pain. They do not address generalized pelvic pain or symptomatology associated with sexual activity. This is not because these questions were not considered in the formulation of the questionnaire. Of 73 questions in the preliminary instrument covering domains of urinary symptoms, pain, sexual function, menstrual variability, and general health, only the four questions now in the instrument were needed to reliably and validly describe the illness experience of those with IC and distinguish these patients from those without the disorder.
| Table 7. O'Leary-Sant Indices. | |||
IC Symptom Index |
IC Problem Index |
||
| During the past month... | During the past month how much has each of the following been a problem for you: | ||
| Q1. ...how often have you felt the strong need to urinate with little or no warning? | Q1. Frequent urination during the day | ||
| 0. Not at all | 0. No problem | ||
| 1. Less than 1 time in 5 | 1. Very small problem | ||
| 2. Less than half the time | 2. Small problem | ||
| 3. About half the time | 3. Medium problem | ||
| 4. More than half the time | 4. Big problem | ||
| 5. Almost always | Q2. Getting up at night to urinate? | ||
| Q2. ...how often have you had to urinate less than 2 hours after you finished urinating? | 0. No problem | ||
| 1. Less than 1 time in 5 | 1. Very small problem | ||
| 2. Less than half the time | 2. Small problem | ||
| 3. About half the time | 3. Medium problem | ||
| 4. More than half the time | 4. Big problem | ||
| 5. Almost always | Q3. Need to urinate with little warning? | ||
| Q3. ...how often did you most typically get up at night to urinate? | 0. No problem | ||
| 0. Not at all | 1. Very small problem | ||
| 2. A few times | 3. Medium problem | ||
| 3. Almost always | 4. Big problem | ||
| 4. Fairly often | Q4. Burning, pain, discomfort, or pressure in your bladder? | ||
| 5. Usually | 0. No problem | ||
| Q4. ...have you experienced pain or burning in your bladder? | 1. Very small problem | ||
| 0. Not at all | 2. Small problem | ||
| 2. A few times | 3. Medium problem | ||
| 3. Almost always | 4. Big problem | ||
| 4. Fairly often | |||
| 5. Usually | |||
| Add the numerical values of the checked entries. | Add the numerical values of the checked entries. | ||
| Total score: ______. | Total score: ______. | ||
The most recently published instrument is the PUF questionnaire (Table 8). It was specifically designed to include questions that directly reflect a wide variety of the symptoms experienced by patients who are affected by this disorder. One third of the questions address pelvic pain, including pain anywhere in the pelvis: the vagina, labia, lower abdomen, urethra, perineum, testes, penis, or scrotum. A large study utilizing the PUF questionnaire has concluded that up to 23% of American females have PBS/IC. This makes one very wary as to the utility and face-validity of the PUF. A total score of 10 to 14 = 74% likelihood of positive potassium test (PST); 15 to 19, 76%; 20+, 91%. To the extent that the PST is suspect, the reliability of PUF data comes into question.
| Table 8. Pelvic Pain and Urgency/Frequency Patient Symptom Scale (PUF). | |||||||||
| PATIENT SYMPTOM SCALE | |||||||||
| Patient's Name: ________________. Today's Date: ___________. | |||||||||
| Please circle the answer that best describes how you feel for each question. | |||||||||
0 |
1 |
2 |
3 |
4 |
Symptom Score | Bother Score | |||
| 1. | How many times do you go to the bathroom during the day? | 3-6 |
7-10 |
11-14 |
15-19 |
20+ |
|||
| 2. | a. | How many times do you go to the bathroom at night? | 0 |
1 |
2 |
3 |
4+ |
||
| b. | If you get up at night to go to the bathroom, does it bother you? | Never bothers |
Occasionally |
Usually |
Always |
||||
| 3. | Are you currently sexually active? |
||||||||
| 4. | a. | If you are sexually active, do you now or have you ever had pain or symptoms during or after sexual activity? | Never |
Occasionally |
Usually |
Always |
|||
| b. | If you have pain, does it make you avoid sexual activity? | Never |
Occasionally |
Usually |
Always |
||||
| 5. | Do you have pain associated with your bladder or in your pelvis (vagina, labia, lower abdomen, urethra, perineum, penis, testes or scrotum)? | Never |
Occasionally |
Usually |
Always |
||||
| 6. | a. | If you have pain, it is usually | Mild |
Moderate |
Severe |
||||
| b. | Does your pain bother you? | Never |
Occasionally |
Usually |
Always |
||||
| 7. | Do you still have urgency after you go to the bathroom? | Never |
Occasionally |
Usually |
Always |
||||
| 8. | a. | If you have urgency, it is usually | Mild |
Moderate |
Severe |
||||
| b. | Does your urgency bother you? | Never |
Occasionally |
Usually |
Always |
||||
| Symptom Score (1, 2a, 4a, 5, 6a, 7, 8a) | |||||||||
| Bother Score (2b, 4b, 6b, 8b) | |||||||||
| Total Score (Symptom Score + Bother Score) | |||||||||
| Total score ranges are from 1 to 35. | |||||||||
The O'Leary-Sant and University of Wisconsin instrument correlates strongly in a large population of patients with PBS/IC. Treatment outcome studies have also used the Global Response Assessment, a balanced patient self-report on overall response to therapy developed for NIDDK-sponsored multicenter therapeutic trials ( Table 9).
| Table 9 . Global Response Assessment (GRA) . |
| - 3: Markedly Worse |
| - 2: Moderately Worse |
| - 1: Slightly Worse |
| 0: No Change |
| +1: Slightly Improved |
| +2: Moderately Improved |
| +3: Markedly Improved |
KEY POINT: CLINICAL SYMPTOM SCALES |
| Symptom scales have potential utility in PBS/IC. Their future development may enable reliable diagnosis of the syndrome on the basis of a questionnaire alone. A brief survey that reliably segregates PBS/IC from other urologic disorders would make the ability to diagnose the syndrome reliable, inexpensive, and available to all health care providers. It would aid in epidemiologic studies as well. Currently, such work sponsored by NIDDK is ongoing. Questionnaires and symptom scales can also be utilized to measure treatment outcome and are especially valuable in clinical research studies as well as for guiding therapy for individual patients. None of the questionnaires has been shown to be of value in diagnosis. |
ASSESSING TREATMENT RESULTS
The diversity of PBS/IC therapies underscores the lack of understanding about the treatment of this syndrome. It has been not only a difficult condition to diagnose but also a difficult condition for which to assess therapeutic impact. There is a 50% incidence of temporary remission unrelated to therapy, with a mean duration of 8 months. A somewhat surprising finding from the ICDB was that although there was initial improvement in symptoms partially due to regression to the mean and the intervention effect, there was no evidence of a long-term change in average symptom severity over the 4-year course of follow-up. In a chronic, devastating condition with primarily subjective symptomatology, no known cause, and no cure, patients are desperate and often seem to respond to any new therapy (Fig. 11). They are often victims of unorthodox health care providers with untested forms of therapy, some medical, some homeopathic, and some even surgical.
|
| Figure 11. Selected reported treatment outcomes in uncontrolled studies in the interstitial cystitis. Percentage of patients initially improved. Ag nitrate, silver nitrate; BCG, bacillus Calmette-Guerin; DMSO, dimethyl sulfoxide; PPS, sodium pentosan polysulfate; TENS, transcutaneous electrical nerve stimulation. |
Placebo effects influence patient outcomes after any treatment, including surgery, in which clinician and patient beliefs are effective. Placebo effects plus disease natural history and regression to the mean can result in high rates of good outcomes, which may be misattributed to specific treatment effects. Unfortunately, few IC treatments have been subjected to a placebo-controlled trial. This is not to say that what seems effective is not but rather that a high index of skepticism is healthy, even in treatments tested by controlled trials. While in many diseases an argument can be made against using a true placebo control as opposed to an orthodox treatment of approved or accepted value, a good case for true placebo can readily be made for IC. The vagaries of the natural history, the general lack of progression of symptom severity over time, and the fact that it is not life threatening mean that there is little to lose and much to gain by subjecting new treatments to the vigorous scrutiny of placebo control. Many patients who volunteer for such trials have already run the gamut of accepted (though generally unproved) therapies. It has long been recognized in protocols that use subjective criteria for assessment that "improvement" may be expected in up to 35% of placebo-treated patients. Because the spontaneous remission rate (though temporary) for IC is 11% to 50%, combined with the placebo improvement it can be difficult to prove efficacy. Even in placebo-controlled trials, it is reasonable to surmise that some degree of unblinding may occur as a result of somatic or psychological side effects of the active arm, impairing the validity of the trial results and giving the active arm a slight edge over placebo. This is of specific concern in PBS/IC and in any disorder in which primary outcomes may be subject to patient-specific psychological and physiologic factors.
The value of placebo-controlled trials is aptly illustrated by the recent decisions by pharmaceutical manufacturers not to pursue U.S. Food and Drug Administration approval for seemingly promising intravesical therapies for PBS/IC after placebo-controlled trials failed to establish efficacy. These include low-concentration hyaluronic acid (Bioniche, Canada), high-concentration hyaluronic acid (SKK, Tokyo), and resiniferatoxin (ICOS, Bothell, WA). Nalmefene, an initially promising oral therapy in the 1990s, also failed phase 3 trials (IVAX, Miami, FL). Placebo trials are impractical in surgery, and it can be difficult to evaluate surgical reports. The many older medications currently used off-label might not meet success if tested in the stringent manner in which new molecular entities are tested. The expense of testing therapies currently used off-label often requires dependence on the largesse of government agencies such as the National Institutes of Health.
Finally, when considering objective changes, the concept of statistical versus clinical significance is paramount. Investigators should, but rarely do, point out differences between statistical improvement and what they consider to be clinically significant improvement). As Gertrude Stein reportedly stated, "A difference, to be a difference, must make a difference". An increase in bladder capacity of 30 mL may be statistically significant but clinically irrelevant. Number needed to treat and number needed to harm data may be particularly important in PBS/IC and have not typically been included in efficacy analysis.
CONSERVATIVE THERAPY
Once the diagnosis has been made one must decide whether to institute therapy or employ a policy of conservative "watchful waiting". If the patient has not had an empirical course of antibiotics for their symptoms by the time the PBS/IC diagnosis is made, such a trial is reasonable. Doxycycline has been reported efficacious in a recent Swiss study. Further attempts to alleviate symptoms with antibiotics are unlikely to be worthwhile and are not recommended in the absence of positive cultures. If the patient's symptoms are "livable", the withholding of immediate treatment is reasonable. While the concept of "livable" is certainly patient dependent, someone who gets up once or twice a night and voids every 2 to 3 hours during the day with minimal pain would certainly fall into this category. An educated patient is likely to realize that no treatment will make him or her perfect, and any therapy is a tradeoff among the inconvenience, chronicity of treatment, side effects, and the benefits. As with any decision in medicine, "perfect is the enemy of good". Data that "early" treatment affects the natural history or course of the disease is lacking at this time, and an argument for the early institution of therapy cannot be supported on the basis of epidemiologic data or clinical trials.
Patient education and empowerment is an important initial step in therapy. Taking advantage of on-line databases, interactive computer programs, electronic mail lists for disabled persons, telephone hotlines, educational videos, health magazines, and public libraries enables patients to make health choices that they can feel comfortable with. It is important to reassure the patient (who often has already seen multiple doctors and had many tests) that the symptoms, although very bothersome, are not signs of a life-threatening disease. Patients are usually reassured after learning that they are not the only persons with these symptoms and that what they have is a part of a well-described syndrome. One must not forget the placebo effect, euphemistically termed "remembered wellness", as one of the physician's most therapeutic assets. The Interstitial Cystitis Association is an important resource for information and support for patients as well as a clearing house for ideas and funding for researchers and clinicians.
There are data that timed voiding and behavioral modification therapy can be helpful in the short-term, especially in patients in whom frequency rather than pain predominates. This can consist of diary keeping, controlled fluid intake, pelvic floor muscle training, and active attempts to increase voiding intervals. Parson's group reported success in 15 of 21 patients who had primarily frequency rather than pain, by gradually encouraging longer voiding intervals over time through voiding diary techniques. A similar study with 15 patients concluded that although average voided volume increased by 65 mL after a month, the persistent sense of bladder fullness did not change from preintervention volumes.
While there are no randomized, controlled trials to prove it, many clinicians believe that stress reduction, exercise, warm tub baths, and efforts by the patient to maintain a normal lifestyle all contribute to overall quality of life. In a controlled study of 45 PBS/IC patients and 31 healthy control subjects, higher levels of stress were related to greater pain and urgency in patients with IC but not in the control group. Maladaptive strategies for coping with stress may impact adversely on symptoms. Biofeedback, soft tissue massage, and other physical therapies may aid in muscle relaxation of the pelvic floor. Mendelowitz and Moldwin had a 69% success rate in 16 patients treated with electromyographic biofeedback, but treatment response did not correlate to changes in muscle identification, and the placebo effect may have been considerable. Acupuncture has been used for PBS/IC and many other chronic pain syndromes. There is limited evidence that it is more effective than no treatment for chronic pain, and there is inconclusive evidence that acupuncture is more effective than placebo, sham acupuncture, or standard care. PBS/IC results with acupuncture have been disappointing.
Elaborate dietary restrictions are unsupported by any literature, but many patients do find their symptoms are adversely affected by specific foods and would do well to avoid them. Often this includes caffeine, alcohol, and beverages that might acidify the urine such as cranberry juice. Anecdotal association of IC with many foods has spawned the recommendation of various "IC diets" with little in the way of objective, scientific basis (Table 10). The only placebo-controlled dietary study, while small, failed to demonstrate a relationship between diet and symptoms. Bade and colleagues found that IC patients tend to have a healthier diet than the general population but could discern no rationale for dietary or fluid intake change other than decreasing caffeine intake. Nguan and coworkers performed a prospective, double-blind, crossover study consisting of crossover instillations of urine at physiologic pH (5.0) and neutral buffered pH (7.5). There was no statistically significant difference in subjective pain scores, suggesting that adjusting urine pH with diet or dietary supplements may have little influence on symptomatology. Orange and grapefruit juices, rich in potassium and citrate, tend to increase urinary pH but surprisingly are avoided by many IC patients based on "IC diet" recommendations. Urinary alkalinization may be worth trying, but supporting studies are lacking.
| Table 10. Interstitial Cystitis Association Recommendations of Foods to Avoid. | |
Milk/Dairy Products |
Nuts |
Aged cheeses Sour cream Yogurt Chocolate Vegetables Fava beans Lima beans Onions Tofu Soybeans Tomatoes Fruits Apples Apricots Avocados Bananas Cantaloupes Citrus fruits Cranberries Grapes Nectarines Peaches Pineapples Plums Pomegranates Rhubarb Strawberries Juices from above fruits Carbohydrates and Grains Rye bread Sourdough bread Meats and Fish Aged, canned, cured, processed, smoked meats and fish |
Beverages Alcoholic beverages including beer and wine Carbonated drinks Coffee Tea Fruit juices Seasonings Mayonnaise Ketchup Mustard Salsa Spicy foods: (Chinese, Mexican, Indian, Thai) Soy sauce Miso Salad dressing Vinegar Preservatives and Additives Benzyl alcohol Citric acid Monosodium glutamate Artificial sweeteners Preservatives Artificial ingredients Food coloring Miscellaneous Tobacco Caffeine Diet pills Junk foods Recreational drugs Allergy medications with ephedrine or pseudoephedrine Certain vitamins |
Unfortunately, education and self-help are often not sufficient, and most patients will require one or more of a variety of therapies.
ORAL THERAPY (Table 11)
Tricyclic Antidepressants
Amitriptyline has become a staple of oral treatment for IC. The tricyclic agents possess varying degrees of at least three major pharmacologic actions: (1) they have central and peripheral anticholinergic actions at some but not all sites, (2) they block the active transport system in the presynaptic nerve ending that is responsible for the reuptake of the released amine neurotransmitters serotonin and noradrenaline, and (3) they are sedatives, an action that occurs presumably on a central basis but perhaps is related to their antihistaminic properties. Amitriptyline, in fact, is one of the most potent tricyclic antidepressants in terms of blocking H1-histaminergic receptors. There is also evidence that they desensitize α2-adrenergic receptors on central noradrenergic neurons. Paradoxically, they also have been shown to block α-adrenergic receptors and serotonin receptors. Theoretically, tricyclic agents have actions that might tend to stimulate predominantly β-adrenergic receptors in bladder body smooth musculature, an action that would further facilitate urine storage by decreasing the excitability of smooth muscle in that area.
| Table 10. Some Oral Medications That Have Been Used for Treatment of PBS/IC. | ||
| Drug | Randomized Control Trial | % Success |
| Amitriptyline | Yes | 42% |
| Antibiotic regimens | Yes | 48% |
| Anticholinergics and antispasmodics | No | Anecdotal |
| Azathioprine | No | 50% |
| Benzydamine | Yes | 0% |
| Chloroquine derivatives | No | 50% |
| Cimetidine | Yes | 65% |
| Cortisone and other steroids | No | 80% |
| Cyclosporine | No | 90% |
| Doxycycline | No | 71% |
| Gabapentin | No | Anecdotal |
| Hormones | No | Anecdotal |
| Hydroxyzine | Yes | 31% |
| L-Arginine | Yes | Not effective |
| Methotrexate | No | 50% |
| Misoprostol | No | 48% |
| Montelukast | No | 90% |
| Nalmefene | Yes | Not effective |
| Narcotic analgesics | No | Anecdotal |
| Nifedipine | No | 87% |
| Phenazopyridine | No | Anecdotal |
| Quercetin | No | 92% |
| Sodium pentosan polysulfate | Yes | 33% |
| Suplatast tosylate | Yes | Pending publication |
| Vitamin E | No | Anecdotal |
Hanno and Wein first reported a therapeutic response in IC after noting a "serendipitous" response to amitriptyline in one of their patients concurrently being treated for depression. The following year a similar report appeared relating a response to desipramine hydrochloride. By reasoning that a drug used successfully at relatively low dosages for many types of chronic pain syndromes, which would also have anticholinergic properties, β-adrenergic bladder effects, sedative characteristics, and strong H1 antihistaminic activity would seem to be ideal for IC, the first clinical trial was carried out with promising results. A subsequent follow-up study reported that in 28 of 43 patients who could tolerate therapy for at least a 3-week trial at a dosage of 25 mg at bedtime gradually increasing to 75 mg at bedtime over 2 weeks, there were 18 who had total remission of symptoms with a mean follow-up of 14.4 months, 5 dropped out because of side effects, and 5 derived no clinical benefit. Benefits were apparent within 4 weeks. All patients had failed hydrodistention and intravesical DMSO therapy. Sedation was the main side effect. Kirkemo and colleagues treated 30 patients and had a 90% subjective improvement rate at 8 weeks. Both studies noted that patients with bladder capacities over 450 to 600 mL under anesthesia seemed to have the best results. Another uncontrolled study of 11 patients with urinary frequency and pelvic pain related success in 9 of the patients, with 5 reporting complete resolution of symptoms and 4 significant relief. Two patients could not tolerate the medication. In a 4-month intent-to-treat, placebo-controlled, double-blind trial of 50 patients, 63% on amitriptyline at doses of 25 to 75 mg (dose as tolerated) before bed reported good or excellent satisfaction versus 4% on placebo. At 19-month follow-up there was little tachyphylaxis and good response rates were observed in the entire spectrum of PBS/IC symptoms.
Amitriptyline has proven analgesic efficacy, with a median preferred dose of 75 mg in a range of 25 to 150 mg daily. This range is lower than traditional doses for depression of 150 to 300 mg. The speed of onset of effect is much faster (1 to 7 days) than reported in depression, and the analgesic effect is distinct from any effect on mood. Tricyclic antidepressants are contraindicated in patients with long QT syndrome or significant conduction system disease (bifascicular or trifascicular block) after recent myocardial infarction (within 6 months), unstable angina, congestive heart failure, frequent premature ventricular contractions, or a history of sustained ventricular arrhythmias. They should be used with caution in patients with orthostatic hypotension. Doses greater than 100 mg are associated with increased relative risk of sudden cardiac death.
Antihistamines
The use of antihistamines goes back to the late 1950s and stems from work by Simmons who postulated that the local release of histamine may be responsible for, or accompany the development of, IC. He reported on six patients treated with pyribenzamine. The results were far from dramatic, with only half of the patients showing some response. The therapy is notable for this disease in that it was very logically conceived. It has been Theoharides who has spearheaded mast cell research in this field and been a major modern proponent of antihistamine therapy. He has used the unique piperazine H1-receptor antagonist hydroxyzine, a first-generation antihistamine, which can block neuronal activation of mast cells. In 40 patients treated with 25 mg before bed increasing over 2 weeks (if sedation was not a problem) to 50 mg at night and 25 mg in the morning, virtually every symptom evaluated improved by 30%. Only 3 patients had absolutely no response. As with many IC drug reports, these responses were evaluated subjectively and without being blinded or placebo controlled. A subsequent study suggested improved efficacy in patients with documented allergies and/or evidence of bladder mast cell activation. No significant response to hydroxyzine was found in an NIDDK placebo-controlled trial. Why an H2 antagonist would be effective is unclear, but uncontrolled studies show improvement of symptoms in two thirds of patients taking cimetidine in divided doses totaling 600 mg. It proved effective in a double-blind, placebo-controlled trial, but histologic studies show the bladder mucosa to be unchanged before and after treatment, and the mechanism of any efficacy remains unexplained.
Sodium Pentosan Polysulfate
Parson's suggestion that a defect in the epithelial permeability barrier, the glycosaminoglycan (GAG) layer, contributes to the pathogenesis of IC has led to an attempt to correct such a defect with the synthetic sulfated polysaccharide sodium pentosan polysulfate (PPS), a heparin analog available in an oral formulation, 3% to 6% of which is excreted into the urine. It is sold under the trade name Elmiron. Studies have been contradictory.
Fritjofsson and associates treated 87 patients in an open multicenter trial in Sweden and Finland. Bladder volume with and without anesthesia was unchanged. Relief of pain was complete in 35% and partial in 23% of patients. Daytime frequency decreased from 16.5 to 13, and nocturia decreased from 4.5 to 3.5. Mean voided volumes increased by almost a tablespoon in the nonulcer group. Holm-Bentzen and colleagues studied 115 patients in a double-blind, placebo-controlled trial. Symptoms, urodynamic parameters, cystoscopic appearance, and mast cell counts were unchanged after 4 months. Bladder capacity under anesthesia increased significantly in the group with mastocytosis, but this had no bearing on symptoms or awake capacity.
Parsons and coworkers had a more encouraging initial experience, and subsequently the results of two placebo-controlled multicenter trials in the United States were published. In the initial study, overall improvement of greater than 25% was reported by 28% of the PPS-treated group versus 13% in the placebo group. In the latter study the respective figures were 32% on drug versus 16% on placebo. Average voided volume on PPS increased by 20 mL. No other objective improvements were documented. An NIDDK 2 ? 2 factorial study to evaluate PPS and hydroxyzine looked at each drug used alone and in combination and compared results with a placebo group. Patients were treated for 6 months. No statistically significant response to either medication was documented. No significant trend was seen in the PPS treatment groups (34%) compared with non-PPS groups (18%). Of the 29 patients on PPS alone, 28% had global response (the primary endpoint) of moderately or markedly improved versus 13% on placebo, a number remarkably similar to the results in the 3-month pivotal trials, although not reaching statistical significance in the 6-month study. A subsequent industry sponsored trial showed no dose-related efficacy response in the range of 300 to 900 mg daily, but adverse events were dose related.
Long-term experience with PPS is consistent with efficacy in a subset of patients that may drop below 30% of those initially treated. Tachyphylaxis seems to be uncommon in responders. Adverse events with PPS occurred in less than 4% of patients at the dose of 100 mg three times daily and include reversible alopecia, diarrhea, nausea, and rash. Rare bleeding problems have been reported. It promotes cellular proliferation in vitro in the MCF-7 breast cancer cell line, and caution has been suggested in prescribing it in groups at high risk for breast cancer and premenopausal females. A 3- to 6-month treatment trial is generally required to see symptom improvement. In a small trial, PPS has shown efficacy when administered intravesically. It may be of value in the management of radiation cystitis and cyclophosphamide cystitis, but its value in the treatment of PBS/IC seems marginal.
Miscellaneous Agents
Several oral therapies for IC have been tried and essentially been discarded, although some are being reinvestigated. Dees reported temporary improvement with systemic corticosteroids, but these drugs have not been found to be useful, and the risks of chronic administration are considerable. Prednisone at a dose of 25 mg daily for 1 to 2 months and then tapered as tolerated may be effective in patients with otherwise unresponsive ulcerative disease. Hormones have been used without success. Vitamin E, anticholinergics, and antispasmodics are not generally efficacious. Immunosuppression and chloroquine derivatives have been administered; about 50% of patients responded to some extent, but potential complications are significant and neither therapy has found general usage. Cyclosporine seemed to have some beneficial effect in a small, uncontrolled study. A follow-up trial showed benefit in 20 of 23 patients treated for a minimum of 1 year. Low-dose oral methotrexate improved bladder pain in 4 of 9 patients and had no effect on voiding pattern. Benzydamine, a potent anti-inflammatory drug with strong analgesic effects, was initially reported to have a superb response rate in IC, but a controlled follow-up trial demonstrated no responses in the first 12 patients, and the trial was discontinued.
The opiate antagonist nalmefene was tried in an uncontrolled trial with promising results. It is known that mast cells degranulate and release histamines and other mediators when their endogenous opioid receptors are stimulated. In PBS/IC this theoretically could occur because of chronic endorphin release stimulated by the pain. By blocking the patient's own endogenous stimulation of mast cell degranulation, a "vicious cycle" could possibly be broken. Unfortunately, an unpublished, placebo-controlled trial failed to demonstrate the hoped-for efficacy.
The calcium channel antagonist nifedipine has been tried in an uncontrolled study for IC and urethral syndrome. Nifedipine has been reported to inhibit detrusor contractions and depress cell-mediated immune functions. Of 9 IC patients treated for at least 4 months, 5 showed a 50% decrease in symptom scores and 33% were asymptomatic. Similar results were noted in patients treated for urethral syndrome. Misoprostol, an oral prostaglandin analog, demonstrated a 48% response in a 9-month open-label trial, although 64% of patients experience adverse drug effects. A cytoprotective action was postulated as the mode of action. The cysteinyl leukotriene D4 receptor antagonist montelukast, a commonly used allergy medication, has shown potential benefits in a small, uncontrolled Danish study.
Oral l-arginine, an over-the-counter amino acid preparation, was purported to increase nitric oxide–related enzymes and metabolites in the urine of IC patients and decrease symptoms, perhaps through relaxation of urinary tract smooth muscle. Later studies showed no change in bladder nitric oxide concentrations after treatment with l-arginine or any significant symptom improvement. An open-label trial of the over-the-counter oral eucalyptus bioflavonoid Quercetin has suggested some efficacy.
Analgesics
The long-term, appropriate use of pain medications forms an integral part of the treatment of a chronic pain condition such as IC. Most patients can be helped markedly with medical pain management using pain medications commonly used for chronic neuropathic pain syndromes including antidepressants, anticonvulsants, and opioids. Many nonopioid analgesics including acetaminophen and the nonsteroidal anti-inflammatory drugs (NSAIDs) and even antispasmodic agents have a place in therapy along with agents designed to specifically treat the disorder itself. Unlike opioids, with increasing doses acetaminophen, aspirin, and the other NSAIDs all reach a ceiling for their maximum analgesic effect. Gabapentin, introduced in 1994 as an anticonvulsant, has found efficacy in neuropathic pain disorders, including diabetic neuropathy and postherpetic neuralgia. It demonstrates synergism with morphine in neuropathic pain. It may give some benefit in chronic pelvic pain syndromes and PBS/IC.
With the results of major surgery anything but certain, the use of long-term opioid therapy in the rare patient who has failed all forms of conservative therapy over many years may also be considered. Opiates are seldom the first choice of analgesics in chronic pain states, but they should not be withheld if less powerful analgesics have failed. This is a difficult decision that requires much thought and discussion between patient and urologist, and involvement of a pain specialist is indicated. A single practitioner has to take responsibility for pain treatment and write all prescriptions for pain medications. Opioids are effective for most forms of moderate and severe pain and have no ceiling effect other than that imposed by adverse effects. The common side effects include sedation, nausea, mild confusion, and pruritus. These are generally transient and easily managed. Respiratory depression is extremely rare if they are used as prescribed. Constipation is common, and a mild laxative is generally necessary. The major impediment to the proper use of these drugs when they are prescribed for long-term nonmalignant pain is the fear of addiction. Studies suggest the risk is low. The long-acting narcotic formulations that result in steady levels of drug over many hours are preferable.
Chronic pain patients often receive inadequate doses of short-acting pain medications, which put them on cycles of short-term relief, anxiety, and pain. It leads to doctor-shopping and drug-seeking behavior confused by physicians with drug addiction. Whereas physical dependence to opioids will be unavoidable, physical addiction, a chronic disorder characterized by the compulsive use of a substance resulting in physical, psychological, or social harm to the user and the continued use despite that harm, is rare. Chronic opioid therapy can be considered as a last resort in selected patients. It is best administered in a pain clinic setting, requiring frequent reassessment by both patient and physician.
INTRAVESICAL AND INTRADETRUSOR THERAPY (Table 12)
Intravesical lavage with one of a variety of preparations has remained a mainstay of treatment in the therapeutic armamentarium of IC. Perhaps the oldest of the intravesical therapies is silver nitrate. The use of silver nitrate has been attributed to Mercier, who reported in 1855 that excellent results with bladder instillations had been obtained in patients suffering from symptoms compatible with IC. Dodson advocated the use of solutions of silver nitrate in increasing strengths as the treatment of choice for this condition. Pool and Rives reported on 74 patients with IC treated with intravesical silver nitrate. The treatment was carried out as follows:
| Table 12. Some Intravesical Medications That Have Been Used for Treatment of PBS/IC. | ||
| Drug | Randomized Controlled Trial | % Success |
| Silver nitrate | No | 60% |
| Clorpactin WCS-90 | No | 60% |
| Dimethylsulfoxide | Yes | 70% |
| Bacillus Calmette-Guerin | Yes | No proven efficacy |
| Resiniferatoxin | Yes | No proven efficacy |
| Hyaluronic acid | Yes | No proven efficacy |
| Heparin | No | 60% |
| Chondroitin sulfate | No | 33% |
| Lidocaine | No | 65% |
| Capsaicin | No | No demonstrated efficacy |
| Oxybutynin | No | Efficacy suggested |
| Doxorubicin | No | Anecdotal efficacy |
| Pentosan polysulfate | Yes | 40% |
A urethral catheter is inserted and the contents of the bladder are evacuated. The bladder is then irrigated with a saturated solution of boric acid. Then 30 to 60 mL of a 1:5000 solution of silver nitrate is instilled into the bladder and permitted to remain there for 3 or 4 minutes if it does not cause intolerable irritation. At the end of this period the solution is permitted to run out through the catheter, which is then withdrawn. The patient usually experiences some dysuria and vesical irritability for 2 or 3 hours. Treatments are repeated every other day. At subsequent treatments, the concentration of silver nitrate in the solution is increased to 1:2500, 1:1000, 1:750, 1:500, 1:400, 1:200, and finally 1:100. If at any time the reaction is too severe, the concentration is increased more slowly.
The initial treatments are performed under general anesthesia, but later treatments are given on an outpatient basis. Ureteral reflux would be a contraindication, and it goes without saying that bladder biopsy would be contraindicated just before instillation for fear of extravasation. Twenty-three years later, Pool wrote that he still considered this treatment regimen the most efficacious form of treatment. He reported excellent results in 70% of patients with a mean response of 7.6 months. Burford and Burford reported a 14% cure rate and 79% improved figure. DeJuana and Everett had a 50% response rate in 102 patients.
O'Connor reported the use of intravesical Clorpactin WCS 90. Clorpactin is a generic term for closely related, highly reactive chemical compositions having a modified derivative of hypochlorous acid in a buffered base. Its activity is dependent on the liberation of hypochlorous acid and its resulting oxidizing effects, wetting and penetrating properties, and detergency. Wishard and associates treated 20 patients with 0.2% Clorpactin gently lavaged in the bladder for 3 to 5 minutes without anesthesia, of whom 14 reported subjective improvement. Murnaghan and coworkers noted improvement in 14 of 17 patients, although 10 required further treatment during the average 2-year follow-up. Most commonly, the treatments are given as described by Messing and Stamey, using 0.4% solution administered at 10 cm H2O under anesthesia. Multiple instillations can be given, with a 1-month pause after the first 2 instillations to await a therapeutic response. Their success rate was 72%, with average 6-month duration of response. La Rock and Sant noted a 50% to 55% meaningful improvement rate occurring within 4 to 6 weeks of treatment. A case of ureteral fibrosis complicating the treatment prompted the recommendation that vesicoureteral reflux be considered a contraindication to the procedure. Our method of Clorpactin delivery is as follows:
Reflux is excluded with a cystogram. Under anesthesia the bladder is distended for 2 minutes at 60 to 80 cm H2O and emptied. The perineum is shielded with a moistened towel. A solution of 0.4% freshly prepared Clorpactin (4 g in 1000 mL of sterile water) is instilled by gravity drainage (the Foley catheter held 10 cm above the level of the bladder) in 150- to 200-mL aliquots for a dwell time of 2 to 3 minutes and drained by gravity. This continues until the entire 1000-mL solution has been used. The bladder and introitus are then irrigated with normal saline, and the catheter is removed.
A mainstay of the treatment of IC is the intravesical instillation of dimethyl sulfoxide (DMSO). DMSO is a product of the wood pulp industry and a derivative of lignin. It has exceptional solvent properties and is freely miscible with water, lipids, and organic agents. Pharmacologic properties include membrane penetration, enhanced drug absorption, anti-inflammatory action, analgesic action, collagen dissolution, muscle relaxation, and mast cell histamine release. In-vitro effects on bladder function belie its positive effects in vivo, where histamine release has not been demonstrated after treatment. It has been suggested that DMSO actually desensitizes nociceptive pathways in the lower urinary tract. Tests for DMSO for treatment of human illness began in the 1960s in the areas of musculoskeletal inflammation and the cutaneous manifestations of scleroderma.
Stewart and colleagues are responsible for popularizing intravesical DMSO for IC. In the mid 1960s they applied it to the skin over the suprapubic area in a group of patients refractory to conventional forms of therapy. Results were poor, but intravesical delivery of 50 mL of a 50% solution instilled for 15 minutes by catheter and repeated at intervals of 2 to 4 weeks showed positive effects in 6 of 8 patients lasting 2 to 12 months. The lack of side effects, other than a garlic-like odor on the breath, or need for inpatient administration were significant breakthroughs over previous treatments. Further reports by this group confirmed safety and efficacy with symptom-free intervals of 1 to 3 months in 73% of patients. Ek and colleagues reported a 70% success rate, but found most patients ultimately required re-treatment or further therapy with other modalities. Prospective series of Fowler and Barker and associates revealed symptomatic success rates of greater than 80%, although relapse was not uncommon. Fowler noted only minimal improvements in functional bladder capacity and attributed the beneficial effects of DMSO to a direct effect on the sensory nerves of the bladder. Perez-Marrero and coworkers compared DMSO to saline and showed a 93% objective improvement and 53% subjective improvement compared with 35% and 18%, respectively, for saline. Patients with bladder instability do not respond.
With its ease of administration, lack of side effects, and dependable symptomatic results, DMSO certainly merits its place as a useful treatment for PBS/IC. I generally administer 50 mL of 50% DMSO as a bladder "cocktail" with 10 mg of triamcinolone, 40,000 units of heparin, and 44 mEq of sodium bicarbonate. In-vivo studies on rat bladder strips exposed to various concentrations of DMSO for 7 minutes showed absence of electrical field stimulation contraction at a 40% concentration and diminished compliance at 30% concentration. Concentrations of 25% or less had negligible effects in this model. How it relates to use of DMSO in humans is unknown. A rare case of eosinophilic cystitis has been reported after DMSO instillation.
Exogenous glycosaminoglycans have been shown to be effective in providing an epithelial permeability barrier in bladders in which the epithelium has been injured with protamine. Heparin, which can mimic the activity of the bladder's own mucopolysaccharide lining, has anti-inflammatory effects as well as actions that inhibit fibroblast proliferation, angiogenesis, and smooth muscle cell proliferation. Because of its numerous effects, the possibility that heparin could be used for therapeutic reasons other than the control of coagulation has been the subject of much inquiry and speculation. Weaver and coworkers first reported intravesical heparin for IC treatment. Given intravesically, there is virtually no systemic absorption, even in an inflamed bladder. While uncontrolled studies suggested some beneficial effect for subcutaneous administration, the obvious risks of anticoagulation and osteoporosis have prevented this form of administration from undergoing further trials and general usage. Ten thousand units can be administered intravesically in sterile water either alone or with DMSO at varying intervals with good results reported. Kuo reported 50% or more improvement in the International Prostate Symptom Score in 29 of 40 women with IC treated with 25,000 units intravesically twice weekly for 3 months. Parsons has used daily intravesical doses of 40,000 units of heparin in 20 mL sterile water administered by the patient daily and held for 30 to 60 minutes. "Reasonable improvement of symptoms" can be expected between 6 months and 2 years after starting therapy. Adding alkalinized lidocaine to the heparin instillation provides better pain relief. These encouraging outcomes must be kept in perspective, given that they are unproven by any placebo-controlled trial.
Another GAG analog, pentosan polysulfate, administered intravesically 300 mg twice weekly in 50 mL of normal saline, showed some modest benefit in a small trial. The nonsulfated GAG, hyaluronic acid, has also been used intravesically. Trials using 40 mg dissolved in 40-mL normal saline weekly for 4 to 6 weeks and then monthly treatments thereafter have had response rates varying from 71% to 30%. In the summer of 2003, Bioniche Life Science and, in the spring of 2004, Seikagaku Corporation reported double-blind, placebo-controlled, multicenter clinical studies of their hyaluronic acid preparations (40 mg/mL or 200 mg/mL, respectively), and neither showed significant efficacy of sodium hyaluronate compared with placebo. These negative studies have not been published in peer-reviewed literature. Neither preparation has been approved for use for PBS/IC in the United States.
Hurst has shown by immunohistochemistry a deficit of chondroitin sulfate from the luminal bladder surface in IC patients. Small uncontrolled studies using intravesical chondroitin sulfate have shown success rates of 33% to 50%.
Doxorubicin and the mast cell stabilizer cromolyn sodium have been tried in pilot trials with the promising results we come to expect in such studies. Follow-up studies are lacking, and these drugs have not become a part of the intravesical pharmacopoeia.
The use of intravesical bacillus Calmette-Guerin (BCG) for IC was first reported by Zeidman and colleagues in 1994 . A subsequent randomized, prospective, double-blind, placebo-controlled trial of 30 patients treated weekly for 6 weeks and followed for a mean of 8 months noted a 60% response rate compared with a 27% placebo response. Surprisingly, BCG was tolerated as well as placebo. Even more surprisingly, 8 of 9 BCG responders continued to have an excellent response in all parameters measured at 27 months of follow-up. It is unclear how BCG achieved this result, but immunologic and/or anti-inflammatory mechanisms have been postulated. A double-blind crossover Swedish study comparing DMSO to BCG failed to substantiate BCG efficacy. A large multicenter randomized controlled trial by NIDDK comparing BCG to placebo found a 12% response rate for placebo compared with a 21% response for BCG. The small response rate failed to reach statistical significance at the P = .05 level, and this large study of 265 patients indicates that BCG has no place in the treatment of moderate to severe PBS/IC. Although the BCG safety profile was considered acceptable in the NIDDK trial, adverse events were not uncommon, and rare hypersensitivity reactions to intravesical BCG can occur.
Efforts to bring new therapies directly to the bladder continue to be the focus of investigators. Oxybutynin has shown efficacy in preliminary studies when administered intravesically at doses of 10 mg dissolved in saline. Electromotive drug administration, the active transport of ionized drugs by the application of an electric current, using lidocaine and dexamethasone, has shown a 25% success rate up to 6 months after instillation. A similar trial using repeated instillations noted success rates of 60% with a mean duration of 6.6 months. Capsaicin, the main pungent ingredient in hot peppers of the genus Capsicum, is a specific neurotoxin that desensitizes C fiber afferent neurons. Resiniferatoxin, an ultrapotent analog of capsaicin, appears to have similar effects with less of the acute pain and irritation associated with capsaicin application. Both compounds have been tested intravesically for the relief of detrusor overactivity. Clinical trials for the use of these compounds in bladder pain and urgency/frequency could show this to be a new and viable treatment modality in the future, but current data on efficacy in IC are lacking. An unpublished phase 2 safety and proof of concept multicenter, placebo-controlled trial conducted by ICOS Corporation of Bothell, Washington, found no significant efficacy of resiniferatoxin compared with placebo, although no safety issues were identified.
The therapeutic value of botulinum toxin type A (BTX-A) stems partially from its ability to temporarily inhibit acetylcholine release and cause flaccid paralysis in a dose-related manner. It can correct focal dystonia when injected into a muscle. In recent years there has been increasing evidence that BTX-A might also have analgesic properties. Initially, this was thought to be due to relief of muscle spasm. However, botulinum has been shown to reduce peripheral sensitization by inhibiting the release of several neuronal signaling markers, including glutamate and substance P, and reducing c-fos gene expression. It may affect the sensory feedback loop to the central nervous system by decreased input from the muscle tissue, possibly by inhibiting acetylcholine release from gamma motor neurons innervating intrafusal fibers of the muscle spindle. BTX-A has been used effectively for years in different conditions with muscular hypercontractions. Intradetrusor BTX administration blocks the acetic acid–induced calcitonin gene-related peptide (CGRP) release from afferent nerve terminals in the bladder mucosal layer in rats. In an animal model of bladder permeability barrier disruption, intradetrusor BTX-A minimized bladder irritability and restored afferent neural responses to baseline levels. These results support clinical trials of BTX-A for the treatment of PBS/IC and other types of visceral pain.
A multi-institutional case series using Botox or Dysport intradetrusor injections in 13 patients with refractory PBS/IC reported improvement in 9 patients. Improvements in symptoms lasted a mean of 3.72 months (range, 1 to 8 months). No systemic complications were observed, although 2 patients had a diminished flow with some need to strain to void. Rackley and colleagues at the Cleveland Clinic reported no change in objective or subjective outcome measures in a series of 10 PBS/IC patients in whom the trigone was spared in the injection technique. At this time, Botox can be recommended for PBS/IC use only in the context of carefully controlled clinical trials.
NEUROMODULATION
As a chronic pain syndrome, it is reasonable to consider therapeutic options that directly interface with the nervous system in the treatment of PBS/IC. This approach is further supported by the association of pelvic floor dysfunction with pelvic pain syndromes.
Pain diversion by transcutaneous electrical nerve stimulation (TENS) is routine in a variety of painful conditions. Fall and colleagues were the first to use electrical stimulation in IC, reporting on 14 patients treated successfully with long-term intravaginal stimulation and TENS. Subsequently, McGuire and associates noted improvement in 5 of 6 patients treated with electrical stimulation.
The primary intention in applying peripheral electrical nerve stimulation in IC is to relieve pain by stimulating myelinated afferents to activate segmental inhibitory circuits. As a secondary effect, urinary frequency may also be reduced. In the most complete review of the subject to date, 33 patients with ulcerative IC and 27 patients with nonulcerative IC were treated by means of suprapubic TENS. Electrodes were positioned 10 to 15 cm apart immediately above the pubic symphysis. High- or low-frequency (2 to 50 Hz) TENS was employed. If there was no effect with high-frequency TENS after 1 month, low-frequency TENS was used. Thirty to 120 minutes of TENS was prescribed daily. Pain improved more than frequency. Good results or remission were described in 26% of nonulcer patients and in a surprising 54% of patients with ulcerative disease. The authors caution that the experience is based on open studies, relatively few patients, and the knowledge of a significant placebo effect with peripheral pain stimulation.
Acupuncture has been used to treat frequency, urgency, and dysuria. Twenty-two of 26 patients treated at the Sp. 6 point had clinically symptomatic improvement. A study looking at both acupuncture and TENS in IC showed limited effects of both modalities. Lumbar epidural blockade is the subject of a positive recent case report, but in an earlier series resulted in only short-term (mean 15 days) pain relief in IC. An Australian double-blind placebo-controlled study of transdermal posterior tibial nerve laser therapy for IC showed no benefit in 56 patients when comparing active to placebo arms, but the placebo effect was remarkably strong, indicating the importance of such trials when evaluating invasive therapies.
Direct sacral nerve stimulation has been explored in the treatment of IC and urgency/frequency and is referred to as neuromodulation, a technique whose urologic potential was developed through the basic and clinical research of Schmidt. He and others have observed that patients who do best with this treatment modality are those who have identifiable pain and dysfunction in the pelvic muscles. Those patients reporting pelvic pain in the absence of demonstrable pelvic floor dysfunction and levator tenderness did poorly. As initially practiced, trial stimulation was performed with a percutaneous temporary electrode for a 3- to 4-day temporary stimulation period to access efficacy. The S3 nerve is most frequently used. A wire electrode is inserted into the foramen and connected to an external pulse generator (Medtronic Inc., Minneapolis, MN). If the trial is successful, the patient would be considered for implantation of a permanent neural prosthesis. More recently, a staged procedure has supplanted the traditional percutaneous approach, because the response to stimulation can be better assessed with more accurate lead placement and stability than through the more hit or miss percutaneous lead placement. Peters' test to implant rate increased from 52% to 94%. Other reports have noted a test to implant rate in the percutaneous technique from 76% in 33 PBS/IC patients to 40% in 211 patients with refractory urge incontinence, urgency-frequency syndrome, and urinary retention.
Neuromodulation has been shown to be effective in treating refractory urinary urge incontinence. Studies on therapeutic potential in PBS/IC followed. The University of Maryland group described decrease in antiproliferative factor activity and normalization of HB-EGF levels in patients with successful test stimulation. Peters and coworkers reported success in two thirds of PBS/IC patients with sacral nerve stimulation. Comiter found 17 of 25 patients were successful with test stimulation and went on to permanent implantation of the Interstim device (Medtronics, St. Paul, MN). Thirteen of 15 who underwent staged implantation were permanently implanted versus 4 of 10 undergoing percutaneous test stimulation. With a mean follow-up of 14 months, 16 of 17 patients were judged successful, giving intent to treat success rate of 64%. Although sacral neuromodulation can decrease narcotic requirements significantly in refractory PBS/IC, the majority of patients taking chronic narcotics for pain will likely continue to use them for pain relief even after implantation.
HYDRODISTENTION
Hydrodistention of the bladder under anesthesia, while technically a surgical treatment, is often the first therapeutic modality employed, often as a part of the diagnostic evaluation. Since there have been no standard methods of distention, results vary markedly. Frontz first suggested hydraulic overdistention of the bladder for IC in 1922, and Bumpus reported the first series 8 years later. Simple bladder filling at cystoscopy will give relief to some patients, whereas Dunn and colleagues reported on 25 patients who underwent distention under anesthesia to the level of the systolic blood pressure for up to 3 hours. Sixteen of the patients were symptom free with a mean follow-up of 14 months; 2 patients suffered bladder rupture. The bladder in IC patients can be very thin, and the possibility of perforation or rupture must always be kept in mind and discussed with the patient. Prolonged distention probably has little or no benefit over a short-term distention measured in minutes. Using epidural anesthesia and a balloon distention technique to the mean arterial pressure for 3 hours continuously, Glemain and colleagues reported good but transient efficacy in patients with a bladder capacity of greater than 150 mL on predistention cystometry. In their prospective series of 30 patients, 18 had maintained a therapeutic response at 6 months and 13 at 1 year of follow-up. Moderate hematuria was almost universal, worsening of symptoms occurred in 5% of patients, and low back and hypogastric pain were common sequelae. There was one bladder rupture, one episode of sepsis, and one episode of prolonged retention.
Our method is to perform an initial cystoscopic examination (which is generally unremarkable), obtain urine for cytology, and distend the bladder for 1 to 2 minutes at a pressure of 80 cm H2O. The bladder is emptied and then refilled to look for glomerulations or ulceration. A therapeutic hydraulic distention follows for another 8 minutes. Biopsy, if indicated, is performed after the second distention. Therapeutic responses in patients with a bladder capacity under anesthesia of less than 600 mL showed 26% with an excellent and 29% with a fair result compared with 12% excellent and 43% fair in patients with larger bladder capacities. Most favorable responses were extremely brief, however, with the exceptional patient noting improvement for 6 months, thus being a candidate for repeat therapeutic distention.
Acute hydrodistention does not seem to result in any long-term bladder dysfunction. Any efficacy is probably related to damage to mucosal afferent nerve endings. It has no benefits in patients with detrusor hyperreflexia or instability. Over half of men with prostate pain and without bacteriuria may have glomerulations. Symptoms in this group have been reported to improve with hydrodistention. While many patients with IC have sensory urgency at awake capacities of less than 100 mL, hydrodistention under anesthesia seems to allow for "staging" of the disease, giving the clinician some idea of the capacity he or she has to work with conservative therapies. A capacity under anesthesia of under 200 mL would not bode well for the likelihood of success of medical therapy. Fortunately, these cases are relatively rare.
SURGICAL THERAPY
The surgical therapy of IC is an option after all trials of conservative treatment have failed, a point that cannot be overemphasized. IC, although a cause of significant morbidity, is a nonmalignant process with a temporary spontaneous remission rate of up to 50%and does not directly result in mortality. Deaths are either self-inflicted or the complications of therapy. Nowhere does the caveat "primum non nocere" bear more relevance; the treatment must be no worse than the disease process. Surgery should be reserved for the motivated and well-informed patient who falls into the category of extremely severe, unresponsive disease, a group that comprises less than 10% of patients.
Many surgical approaches have been employed for IC, and it is worth mentioning a few for historical perspective alone. Sympathectomy and intraspinal alcohol injections have been used to treat pelvic pain. Differential sacral neurotomy was reported in 3 patients with good results but, like most denervation procedures, never gained popularity because of subsequent poor results. Transvesical infiltration of the pelvic plexuses with phenol failed in 5 of 5 patients with IC. With a significant complication rate of 17% it is rarely used. There are several reports on cystolysis going back to Richer in 1929. Worth and Turner-Warwick reported some short-term benefit, but unpredictable long-term results. Freiha and Stamey used it in 6 IC patients with good results in 4. Albers and Geyer reported long-term follow-up in 11 IC patients and had only one success. Denervation procedures have a notoriously high late-failure rate, and the procedure is not justified for IC. In fact, Rogers has concluded that there exist no convincing clinical studies to recommend surgical procedures to interrupt visceral nerve pathways in women suffering with any type of chronic pelvic pain.
Transurethral resection of a Hunner's ulcer, as initially reported by Kerr , can provide symptomatic relief. Fall resected ulcerated lesions in 30 patients resulting in initial disappearance of pain in all and a decrease in urinary frequency in 21. Similar results have been attained with the neodymium:yttrium-aluminum-garnet laser. Extreme caution is critical if using a laser in an IC bladder, because forward scatter through these thin bladders with resulting bowel injury is an ever-present danger. There would seem to be no justification in the literature for using the laser to treat areas of glomerulation or in the nonulcerative form of the disease.
Supratrigonal cystectomy and the formation of an enterovesical anastomosis with bowel segments has been a popular surgical procedure for intractable IC. The diseased bladder is resected in its entirety, sparing only a 1-cm cuff around the trigone to which the bowel segment is anastomosed. While it is not always clear in the literature how much bladder has been resected, the results reported using these procedures for IC have been mixed at best. Badenoch operated on 9 patients, with 4 becoming much worse and 3 ultimately undergoing urinary diversion. Flood and colleagues reviewed 122 augmentation procedures, 21 of which were done for IC. Patients with IC had the poorest results of any group, with only 10 having an "excellent" outcome. Wallack and colleagues reported two successes; Seddon and associates had success in 7 of 9 patients; and Freiha and coworkers ended up performing formal urinary diversion in 2 of 6 patients treated with augmentation cecocystoplasty. Weiss and associates had success in 3 of 7 patient treated with sigmoidocystoplasty, and Lunghi and colleagues had no excellent results in 2 patients with IC. Webster and Maggio reviewed their data in 19 patients and concluded that only patients with bladder capacities under anesthesia less than 350 mL should undergo substitution cystoplasty. Hughes and associates lowered the threshold to less than 250 mL.
More recent series on subtotal cystectomy plus augmentation have been somewhat more positive. Peeker and associates had good results in all 10 patients with ulcerative IC but poor results in the 3 patients operated on with nonulcerative disease. They no longer perform the procedure in the latter group. Linn and colleagues had success in 20 of 23 patients (only 2 with ulcerative IC) treated with subtotal cystectomy and orthotopic bladder substitution with an ileocecal pouch. They recommend a supratrigonal cystectomy. A Spanish series reported success in 13 of 17 procedures with a mean follow-up of 94 months. The University of Alabama group reported long-term success in 1 of 4 patients with orthotopic neobladders and 1 of 3 with augmentation cystoplasty. A German report on substitution cystoplasty sparing the trigone was quite enthusiastic, detailing a 78% pain-free rate in 18 patients treated with ileocecal augmentation (10) or ileal substitution (8) at a mean follow-up of 57 months. Two patients failed to get any pain relief, and 4 required either long-term intermittent catheterization or suprapubic drainage to empty the neobladder.
Not all patients empty the bladder spontaneously after substitution cystoplasty. Although the need for clean intermittent catheterization would not obviate a successful outcome in the patient treated for bladder contraction from tuberculous cystitis, it can be a painful disaster in the IC patient. Nurse and colleagues have gone one step farther, recommending trigone biopsy before substitution cystoplasty. Diversion and/or total cystourethrectomy is recommended if the trigone is "affected" by IC. It is not clear how this is determined histologically, because IC has no pathognomonic findings by histology and generally is not a localized process. Nielsen and coworkers described 8 women treated with substitution cystoplasty. In 6 patients the procedure failed, and the results of postoperative biopsies from the trigone showed no difference in the amount of fibrosis, degree of degenerative changes in the muscle, and mast cell density between the 2 cured patients and the others.
There has been a controversy over whether the IC process can occur in a transposed bowel patch, or even in the ureter. If so, not only would this be a relative contraindication to the procedure, but it would also provide support for the view that a substance in the urine might be involved in pathogenesis. There is, however, evidence that inflammation and fibrosis are the usual reactions of bowel to exposure to urine and, therefore, pathologic findings alone would not be conclusive of spread of IC in those patients.
Augmentation cystoplasty has many potential complications from the rare incidence of bladder neoplasm to the more common complication of upper tract obstruction. In the best of hands complications can involve almost 50% of patients, requiring surgical intervention in 25%. Whereas problems are more common in patients operated on for disorders other than IC, the risk-benefit ratio of substitution cystoplasty seems to have discouraged its use in the last several years.
Urinary diversion with or without cystourethrectomy is the ultimate surgical answer to the dilemma of IC, akin to cutting the "Gordian knot". If diversion alone is chosen, one must keep in mind potential problems that can befall the remaining bladder, including pyocystitis, hemorrhage, severe pain, and unremitting feelings of incomplete emptying and spasm. Bladder carcinoma has also been reported after urinary diversion but is not specifically associated with IC. Cystourethrectomy is certainly indicated in patients who are miserable and have not only failed all other therapies but have demonstrated chronicity such that remission is considered extremely unlikely. Fortunately few patients fall into this category. Theoretically, conduit diversion seems to be reasonable if one is concerned about disease occurring in any continent storage type of reconstruction. The extended simple cystectomy performed for intractable IC may lend itself to anterior enterocele formation from weakening of the anterior vaginal wall, and prevention of this entity is warranted at the time of cystectomy.
Bejany and Politano reported excellent results in 5 patients treated with total bladder replacement and recommend neobladder reconstruction. Keselman and associates had 2 failures in 11 patients treated with continent diversion and attributed this to surgical complications. A Finnish group noted failure in 2 of 4 patients treated with cystectomy and conduit diversion because of persistent pain. Baskin and Tanagho also cautioned about persistence of pelvic pain after cystectomy and continent diversion, discussing 3 such patients. A similar report followed. Webster and coworkers had 10 failures in 14 patients treated with urinary diversion and cystectourethrectomy. Ten patients had persistent pelvic pain, and 4 of them also complained of pouch pain. Only two patients had symptom resolution. An English study of 27 patients who underwent cystectomy and bladder replacement with a Kock pouch noted successful treatment of pain in all patients, but follow-up was limited. Parsons suggests that pouch pain will occur in 40% to 50% of patients within 6 to 36 months of surgery.
Attempts have been made to improve results by limiting the operation to those without detrusor mastocytosis and those without "neuropathic pelvic pain". Based on the experience of the past decades, it is unclear if these efforts will prove any more successful. It would seem that risks of failure peculiar to IC include both the development of pain over time in any continent storage mechanism that is constructed and the risk of phantom pain in the pelvis that persists despite the fact that the stimulus that initially activated the nociceptive neurons (diseased bladder) has been removed. Brookhoff has proposed trying a differential spinal anesthetic block before considering cystectomy. If the patient continues to perceive bladder pain after a spinal anesthetic at the T10 level, it can be taken as an indicator that the pain signal is being generated at a higher level in the spinal cord and that surgery on the bladder will not result in pain relief. Some patients with intractable urinary frequency will opt for simple conduit urinary diversion alone, feeling that their quality of life will be improved independent of the pain piece of the puzzle. Despite all of the problems, many patients will do well after major surgery and quality of life can measurably improve. In the event of neobladder pain after subtotal cystectomy and enterocystoplasty or continent diversion, it appears safe to retubularize a previously used bowel segment to form a urinary conduit for a straightforward urinary diversion without significant risk of conduit pain.
Forty years ago Pool recognized that "surgical treatment has not been the boon many had hoped it would be". "Diversion of the urine is not the entire answer to the situation. Removal of the lesion in the bladder has been of no benefit. Likewise, removal of almost the entire mobile portion of the bladder proved to be a failure". Blaivas and colleagues described results of augmentation enterocystoplasty and continent diversion in 76 consecutive patients with benign disease with a mean 9-year follow-up. All 7 patients with the diagnosis of IC were classified as failures whereas 67 of the remaining 69 patients were cured or improved. When one of the deans of major urologic reconstruction writes, "I find it very difficult to justify such extensive surgery (continent diversion, cystourethrectomy) with such limited results and for these reasons have not been involved in surgery for IC over the past 3 years", it is obvious that one should think carefully and proceed with surgery only after a complete discussion with a very motivated and well-informed patient.
|
| Figure 12. Suggested diagnosis and treatment algorithm for painful bladder syndrome. |
KEY POINTS: PRINCIPLES OF MANAGEMENT |
|
URETHRAL SYNDROME
In 1945, the distinguished American physician Richard Cabot was quoted as having stated that "any pain within two feet of the female urethra for which one cannot find an adequate explanation should be suspected of coming from the female urethra". The term urethral syndrome was first mentioned in a clinicopathologic study of the female urethra in 1949. It appeared in the British literature in 1965 when a group of New Zealand physicians used it to describe the 50% of their female patients with urinary symptoms without demonstrable infection. The urethral syndrome is a very nonspecific constellation of symptoms including urinary frequency, urgency, dysuria, and suprapubic discomfort without any objective findings of urologic abnormality to account for the symptoms. Although the symptoms are typically thought to occur in women, there is no reason to assume that a similar entity does not occur in men. The frequency, urgency, and suprapubic, perineal, and low back pain of the chronic pelvic pain syndrome in men are certainly reminiscent of the urethral syndrome.
The urethral syndrome has been subdivided into an acute and a chronic condition. In the past, dysuric women with midstream urine cultures containing 105 bacteria/mL or greater were usually considered to have cystitis and those with lower-count bacterial cultures were said to have the urethral syndrome. Since about 1980, it has become apparent that infections of the urethra, urinary bladder, and vagina account for a large proportion of patients who develop the symptom-complex acutely. Therefore, the term acute urethral syndrome, implying as it does a mysterious cause and a urethral origin of the malady, has largely been abandoned in favor of identifiable etiologic diagnoses , many of which are found in the chapters on urinary tract infection and sexually transmitted diseases. Only a relatively small percentage of patients with acute urethral syndrome are found on investigation to have no cause for the symptoms. It would be more accurate to categorize this group of patients by their symptoms than to give them a diagnosis of "acute urethral syndrome", which ultimately communicates little about the disorder.
Those patients with chronic symptoms and with no apparent cause constitute the chronic urethral syndrome category. This phantom diagnosis is one of exclusion and is rarely used in modern urologic texts. The symptomatic manifestations of IC and the chronic urethral syndrome are indistinguishable.
The concept of the urethral syndrome, chronic or acute, is now essentially a historical one and no longer alluded to in the modern medical literature. Some of the many causes of urinary frequency and urgency are listed in Table 13 .
| Table 13. Causes of Frequency and Urgency. |
|
